Bare Metal Coronary Stents
Price 200 USD ($)/ Piece
Bare Metal Coronary Stents Specification
- Usage Type
- Single Use
- Accuracy
- Precise Deployment
- Function
- Restores Coronary Blood Flow
- Features
- High Flexibility, High Radial Strength, Thin Strut Design
- Instruments Type
- Interventional Cardiology Device
- Storage Instructions
- Store in Dry, Room Temperature, Away from Sunlight
- Measurement Range
- 2.0 mm to 4.0 mm diameter, 8 mm to 38 mm length
- Shelf Life
- Up to 3 Years
- Equipment Type
- Bare Metal Coronary Stents
- Material
- Cobalt Chromium / Stainless Steel
- Condition
- New
- Technology
- Laser-Cut, Balloon Expandable
- Portable
- Yes
- Wall Mounted
- No
- Real-Time Operation
- Yes
- Noise Level
- Silent
- Operating Type
- Manual Deployment
- Use
- Coronary Artery Disease Treatment
- Dimension (L*W*H)
- Varies (e.g., 8-38 mm length, 2.0-4.0 mm diameter)
- Weight
- Ultra-lightweight (a few milligrams)
- Color
- Metallic Silver
- Packaging
- Sterile Blister Pack
- Latex Content
- Latex-free
- Radiopacity
- High (visible under fluoroscopy)
- Stent Design
- Open and Closed Cell Design Options
- Endothelialization
- Promotes Fast Vessel Healing
- Delivery System Compatibility
- 6F or 5F Guiding Catheters
- Expansion Capability
- Over-expansion up to 0.5 mm
- Crossability
- Superior, for tortuous vessels
- CE Marked
- Yes
- Strut Thickness
- 60-100 microns
- Sterilization
- Ethylene Oxide Sterilized
- Surface Finish
- Electropolished
About Bare Metal Coronary Stents
Advin Health Care offers Bare Metal Coronary Stents (BMS) designed to meet international quality standards. These stents provide effective mechanical scaffolding to coronary arteries, restoring vessel patency and ensuring improved blood flow in patients with coronary artery disease, without the use of drug coatings.
Key Features
- Reliable mechanical support for coronary arteries
- High radial strength for optimal vessel expansion
- Smooth surface finish for controlled deployment
- Excellent flexibility for navigation in tortuous vessels
- Biocompatible materials for safe and effective use
Specifications
- Type: Bare Metal Coronary Stent (BMS)
- Material: Stainless Steel / Cobalt Chromium
- Coating: None (Non-drug eluting)
- Delivery System: Balloon-expandable system
- Sterilization: EO Sterilized
- Usage: Single-use
- Application: Treatment of coronary artery disease (CAD)
Certifications
CE, ISO 13485, and ISO 9001 certified
Product Keywords
Bare Metal Coronary Stent, BMS Stent, Coronary Stent, Cardiac Stent, Angioplasty Stent, Non-Drug Eluting Stent
BMS
, , , , , .
Stent coronario de metal desnudo, stent BMS, stent coronario, stent cardaco, stent de angioplastia, stent no liberador de frmacos
Stent coronaire nu, stent BMS, stent coronaire, stent cardiaque, stent d'angioplastie, stent sans lution mdicamenteuse
Advanced Design for Enhanced Performance
The stents feature thin struts and an electropolished finish, resulting in high flexibility and radial strength. With both open and closed cell architectures, these devices ensure tailored support according to vessel anatomy and interventional needs. Their ultra-lightweight structure allows for superior crossability in complex, tortuous vascular anatomies.
Safe and Sterile Packaging
Each stent is supplied in a sterile blister pack and sterilized using ethylene oxide, ensuring patient safety. The latex-free composition eliminates the risk of allergic reactions, and all products are CE Marked, adhering to stringent international medical standards.
Compatibility and Ease of Use
Designed for compatibility with 5F and 6F guiding catheters, these stents offer precise, manual balloon expansion. Their wide range of diameters (2.0-4.0 mm) and lengths (8-38 mm) accommodates various patient requirements, while ensuring accurate and efficient deployment during coronary interventions.
FAQ's of Bare Metal Coronary Stents:
Q: How are these bare metal coronary stents deployed during a procedure?
A: These stents are balloon-expandable and deployed manually using compatible 5F or 6F guiding catheters. The stent is advanced across the lesion using a delivery system, then expanded by inflating the balloon, ensuring precise positioning and effective vessel support.Q: What benefits do thin strut designs and electropolished surfaces provide?
A: Thin strut designs (60-100 microns) and electropolished surfaces increase flexibility, improve crossability, and help the stent conform to tortuous vessels. They also promote rapid endothelialization, leading to faster vessel healing and reducing the risk of restenosis.Q: When should a clinician choose an open versus closed cell stent design?
A: Open cell designs provide enhanced flexibility and conformability, making them ideal for tortuous or branching vessels. Closed cell designs offer greater radial strength and uniform scaffolding, preferred in straight or heavily calcified arteries.Q: Where should these bare metal stents be stored before use?
A: Stents should be stored in their original sterile blister pack, in a dry and room temperature environment, away from direct sunlight. This ensures product integrity and maintains a shelf life of up to 3 years.Q: What is the measurement range available for length and diameter of these stents?
A: These stents are available in lengths ranging from 8 mm to 38 mm and diameters from 2.0 mm to 4.0 mm, accommodating a wide variety of vessel sizes during coronary interventions.Q: How does radiopacity benefit interventional cardiologists during procedures?
A: High radiopacity ensures that the stent is clearly visible under fluoroscopy, allowing interventional cardiologists to track and position the stent accurately within the coronary arteries, thereby improving procedural safety and outcomes.Q: What makes these stents suitable for export and supply in India and globally?
A: These stents meet CE Mark standards and employ advanced laser-cut, balloon-expandable technology. Their sterile, secure packaging and high safety profile make them suitable for hospitals, clinics, and medical suppliers in India and international markets.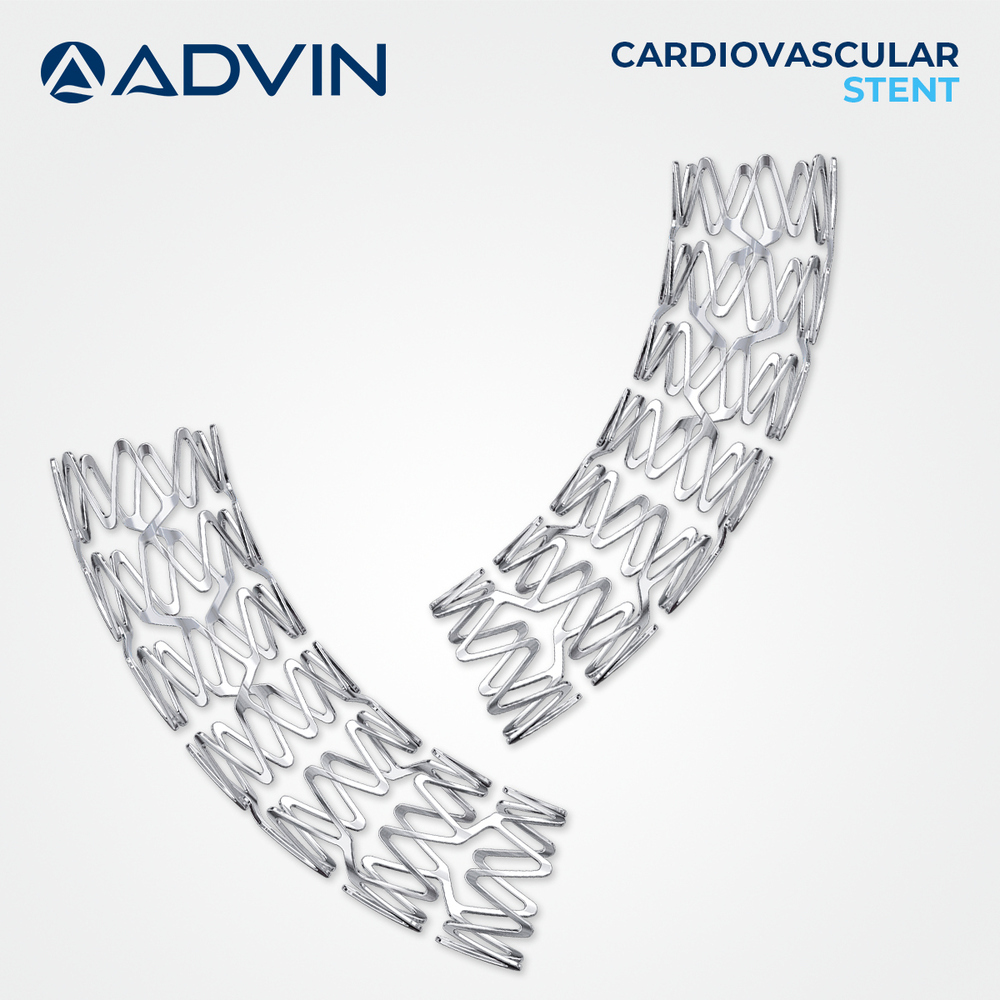




Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Cardiology Products Category
PVC Surgical Equipments Hybrid Type Inflation Device
Price 20 USD ($) / Piece
Minimum Order Quantity : 1 Piece

 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry

 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese