Cardiac Pacing Leads
Price 30 USD ($)/ Piece
Cardiac Pacing Leads Specification
- Function
- Delivers Electrical Impulse to Stimulate Cardiac Muscle
- Usage Type
- Disposable, Single-use
- Storage Instructions
- Store in a Cool, Dry Place; Avoid Direct Sunlight
- Shelf Life
- 2-3 Years
- Features
- Flexible, Non-thrombogenic, Kink-resistant, Radiopaque Tip, Sterile Packaging
- Accuracy
- High Pacing Accuracy
- Instruments Type
- Cardiac Electrophysiology Device
- Equipment Type
- Cardiac Pacing Leads
- Material
- Biocompatible Polymer and Metal Alloy
- Condition
- New
- Technology
- Transvenous Pacing Technology
- Portable
- Yes
- Wall Mounted
- No
- Real-Time Operation
- Yes
- Noise Level
- Low
- Operating Type
- Manual & Compatible with External Pacemakers
- Use
- Temporary Cardiac Pacing
- Power Source
- External Pacemaker (Not Included)
- Dimension (L*W*H)
- Varies (Usually Length: 50-100 cm, Diameter: 6-8 Fr)
- Weight
- Lightweight (< 50g per lead)
- Color
- White/Clear with Metallic Tip
- Patient Compatibility
- Pediatric and Adult
- Radiopacity
- Radiopaque Tip for Visualization under Fluoroscopy
- Tip Configuration
- Straight or Curved
- Sterilization Mode
- Ethylene Oxide (EtO) Sterilized
- Catheter Size
- 6 Fr, 7 Fr, 8 Fr
- Latex-Free
- Yes
- Lead Insulation
- Silicone or Polyurethane Coated
- Regulatory Approval
- CE Marked, ISO 13485 Certified
- Connector Type
- IS-1 or Compatible Pin Connector
- Flexibility
- High Flexibility for Easy Navigation
- Packaging Type
- Individual Sterile Blister Pack
- Length Options
- Available in Multiple Lengths
About Cardiac Pacing Leads
Advin Health Care offers Cardiac Pacing Leads designed to meet international quality standards. These leads are used to deliver controlled electrical impulses to the heart for maintaining or restoring normal cardiac rhythm. Suitable for temporary pacing applications, they ensure reliable performance in ICU, emergency, catheterization lab, and surgical environments.
Key Features
- Reliable electrical conduction for consistent pacing output
- Flexible and kink-resistant design for smooth navigation
- Atraumatic tip for safe and comfortable placement
- Stable positioning for effective myocardial contact
- Compatible with standard external pacemakers
- Available in multiple tip configurations for clinical flexibility
Specifications
- Type: Cardiac Pacing Lead
- Material: Medical-grade polymer with conductive core
- Variants: Normal Tip / J-Tip / Balloon Tip / Myocardial (Epicardial)
- Sterilization: EO Sterilized
- Usage: Single-use / Disposable
- Application: Temporary cardiac pacing in ICU, emergency, and surgical procedures
Certifications
CE, ISO 13485, and ISO 9001 certified
Product Keywords
Cardiac Pacing Lead, Temporary Pacing Lead, Cardiac Pacing Wire, Transvenous Pacing Lead, ICU Pacing Lead
, , , , .
Cable de marcapasos cardaco, cable de marcapasos temporal, cable de marcapasos cardaco, cable de marcapasos transvenoso, cable de marcapasos para UCI
Sonde de stimulation cardiaque, sonde de stimulation temporaire, fil de stimulation cardiaque, sonde de stimulation transveineuse, sonde de stimulation pour soins intensifs
Advanced Design for Precision and Safety
These cardiac pacing leads combine biocompatible polymer and metal alloy construction for enhanced patient safety and high electrical performance. The radiopaque tip assists clinicians in accurate placement, while the choice of straight or curved configurations provides flexibility for diverse anatomical requirements.
Effortless Placement and Reliable Operation
Their flexible, kink-resistant body allows smooth transvenous navigation, reducing complications during temporary pacing procedures. The leads are compatible with most external pacemaker units, supporting real-time operation with minimal noise and high efficiency. Each lead is single-use and individually sterile-packed for optimal infection control.
Stringent Quality and Compliance Standards
Every lead is CE marked and produced under ISO 13485-certified manufacturing, reflecting strict quality control and assurance. These devices are latex-free, suitable for a wide spectrum of patients, and come with a shelf life of 2-3 years, ensuring reliability and readiness whenever needed.
FAQ's of Cardiac Pacing Leads:
Q: How are the cardiac pacing leads sterilized before use?
A: These cardiac pacing leads are sterilized using ethylene oxide (EtO) gas, ensuring they are safe and free from contaminants prior to clinical use. Each lead is packaged in an individual sterile blister pack to maintain its integrity until needed.Q: What are the benefits of the radiopaque tip on the pacing leads?
A: The radiopaque tip enhances visualization under fluoroscopy, allowing clinicians to accurately place the lead within the heart. This feature supports precise positioning and improves patient safety during the temporary pacing procedure.Q: When should a straight tip versus a curved tip configuration be selected?
A: The choice between straight and curved tip configurations depends on patient anatomy and physician preference. Curved tips can assist in navigating challenging vascular paths, while straight tips are ideal for more straightforward anatomical routes.Q: Where can these cardiac pacing leads be used?
A: These leads are designed for use in hospitals, cardiac care units, and electrophysiology labs, wherever temporary cardiac pacing is required. They are compatible with a wide range of external pacemakers, supporting both pediatric and adult patient populations.Q: What is the process for storing unused cardiac pacing leads?
A: Unused leads should be stored in their original, sealed, sterile blister packaging in a cool, dry place, away from direct sunlight, to maintain sterility and product longevity. Proper storage extends shelf life up to 2-3 years.Q: How do these leads ensure flexibility and kink resistance during placement?
A: These cardiac pacing leads are constructed with durable silicone or polyurethane insulation and designed with a flexible, non-thrombogenic body. This allows for easy navigation through veins and minimizes the risk of kinking during the procedure.Q: What is the advantage of high pacing accuracy in these leads?
A: High pacing accuracy ensures reliable and consistent electrical stimulation of the cardiac muscle, optimizing therapeutic outcomes and supporting patient safety during temporary pacing.



Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Cardiology Products Category
Cardiac surgery medical kit
Price 6.00 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Technology : Precision Manufacturing, Sterile Packaging
Instruments Type : Surgical Instruments (Forceps, Scissors, Clamps, Needle Holders, Retractors, etc.)
Shelf Life : Up to 5 Years (Unopened and Stored Properly)
Features : Sterile, Disposable Options, Complete Set for Adult Cardiac Surgery
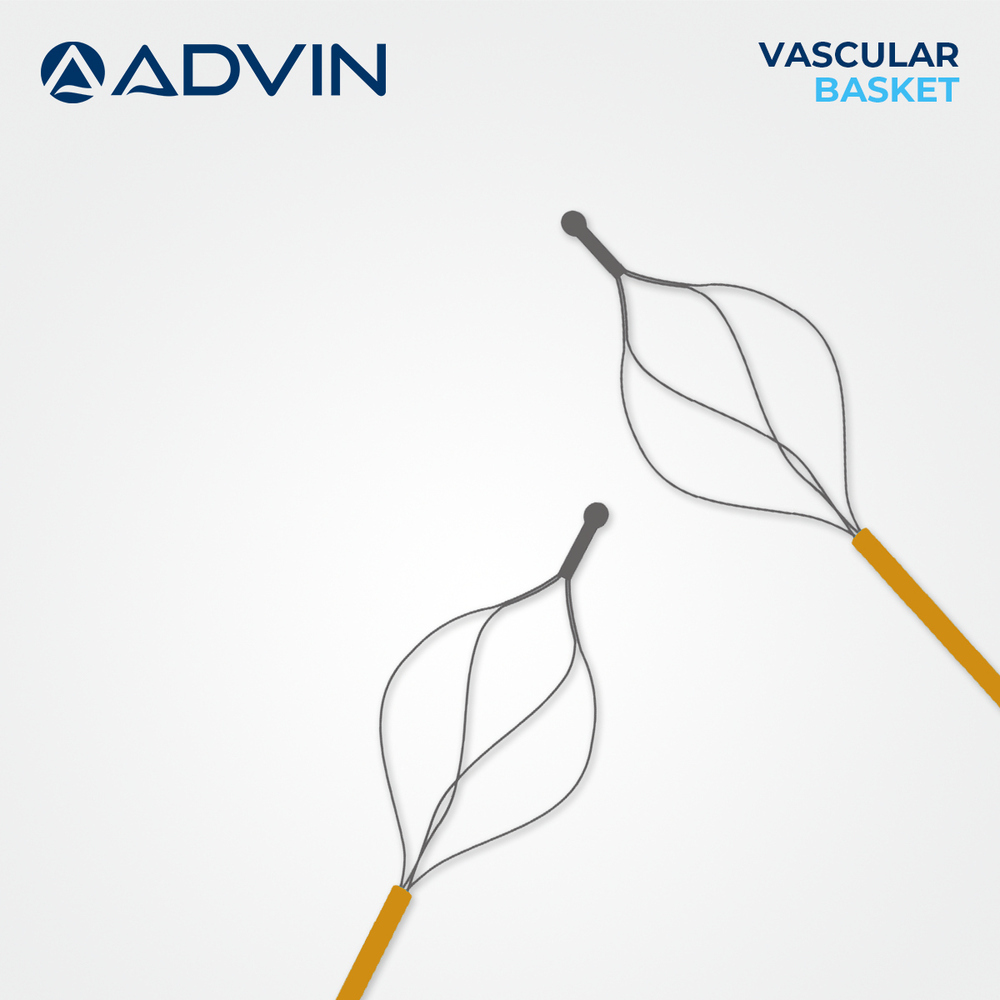
Clinical Vascular Snare Basket
Price 70 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Technology : Advanced Vascular Retrieval
Instruments Type : Interventional Device
Shelf Life : 3 Years
Features : Radiopaque Marker, Flexible Design, High Retention Strength
Angioway High Pressure Extension Tubings
Price 0.50 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Technology : Pressureresistant tubing technology
Instruments Type : Disposable Medical Tubing
Shelf Life : 5 Years
Features : High pressure resistance, DEHP free, leakage proof, flexible, kink resistant, sterile packaging

 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry


 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese