Coronary Artery Stent
Price 200 USD ($)/ Piece
Coronary Artery Stent Specification
- Measurement Range
- Diameter: 2.5-4.0 mm; Length: 8-38 mm
- Features
- Biocompatible, Radiopaque, Flexible
- Usage Type
- Single Use
- Shelf Life
- 3 Years
- Accuracy
- High placement precision
- Instruments Type
- Interventional Cardiology Device
- Storage Instructions
- Store in a dry place below 30C
- Function
- Restores blood flow by scaffolding artery
- Equipment Type
- Coronary Artery Stent
- Material
- Cobalt Chromium Alloy
- Condition
- New
- Technology
- Drug Eluting Stent (DES)
- Portable
- Yes
- Wall Mounted
- No
- Real-Time Operation
- Yes
- Noise Level
- Silent
- Operating Type
- Manual Placement (via Catheter)
- Use
- Coronary Artery Disease Treatment
- Dimension (L*W*H)
- Length: 8-38 mm, Diameter: 2.5-4.0 mm
- Weight
- Negligible/lightweight
- Color
- Metallic (Silver-Grey)
- Strut Thickness
- 80-120 m (microns)
- Recoil
- Low recoil design
- Surface Finish
- Electropolished Smooth Surface
- Delivery System
- Pre-mounted on delivery balloon catheter
- Expansion Method
- Balloon expandable
- Radiopacity
- Yes (visible under fluoroscopy)
- Intended Population
- Adults with coronary artery disease
- Sterility
- Sterile, EO Gas/ Gamma Irradiation
- Compatibility
- Compatible with 0.014" guidewire
- Coating Type
- Polymer-based (Drug: Sirolimus or Everolimus)
- Packaging
- Individually packed, sterile blister pack
About Coronary Artery Stent
The coronary artery stent is designed to provide reliable vessel support following angioplasty procedures. Its structure helps maintain arterial patency and supports normal blood flow to the heart muscle. Advin Health Care manufactures this stent with a strong focus on clinical safety, durability, and consistent performance. The stent is widely used in the treatment of coronary artery disease and supports effective long-term outcomes in cardiac care.
Advanced Features- Designed to provide stable vessel support after angioplasty
-
Helps maintain coronary artery patency
-
Compatible with standard stent delivery systems
-
Intended for use in coronary intervention procedures
- Certifications
We are certified with CE, ISO 13485, and ISO 9001 standards.
Product Keywords
Coronary Artery Stent, Coronary Stent, PCI Stent, Angioplasty Stent, Interventional Cardiology Stent, Drug-Eluting Stent, Bare Metal Stent
1U U UU UaU 1U U UU UaU 1U UU1U UaU 1U UUU Ua UU 1U UUU U UU 1U U1 UU oU UUU
N12N N o34N3412N12341 NNN , o34N3412N12N1 NN12N, NN12N N , NN12N N 123 34NN o , NN12N N 12NN212N 341212341 oN 34343 , NN12N N oNNN21212N14 34oNNN 14, NN12N 12N2NN1 NN
Stent de arteria coronaria, stent coronario, stent para ICP, stent para angioplastia, stent para cardiologAa intervencionista, stent liberador de fArmacos, stent metAlico desnudo
Stent coronaire, stent d'angioplastie, stent pour cardiologie interventionnelle, stent A Alution de mAdicament, stent nu
Advanced Drug Eluting Stent for Precise Coronary Intervention
Our coronary artery stent incorporates a low-recoil, flexible design to support vessel healing and reduce the risk of restenosis. The electropolished surface ensures smooth arterial contact, while polymer-based coatings release Sirolimus or Everolimus to inhibit abnormal tissue growth. Size versatility caters to various lesion lengths and vessel diameters, enhancing its applicability across diverse cardiac interventions. Radiopaque features facilitate precise placement under real-time fluoroscopy for optimal outcomes.
Sterile and Secure Packaging for Safe Handling
Each stent is delivered pre-mounted on its balloon catheter and sealed in an individually sterile blister pack, ensuring protection against contamination. The product undergoes either EO gas or Gamma irradiation sterilization to meet stringent safety protocols. Shelf life extends up to three years when stored below 30C in a dry environment, guaranteeing reliability whenever the need arises.
FAQ's of Coronary Artery Stent:
Q: How is this coronary artery stent delivered and implanted?
A: The stent comes pre-mounted on a balloon catheter and is designed for manual placement by an interventional cardiologist. It is navigated to the lesion using a 0.014" guidewire and expanded using balloon inflation under real-time fluoroscopy to ensure accurate positioning within the artery.Q: What are the benefits of the electropolished surface and polymer-based drug coating?
A: The electropolished surface minimizes friction, allowing smoother vessel navigation and reducing vascular trauma. The polymer-based coating releases Sirolimus or Everolimus, drugs known to suppress excessive cell growth, lowering the risk of restenosis and promoting long-term vessel patency.Q: When should this stent be used in clinical practice?
A: This stent is specifically intended for adults diagnosed with coronary artery disease who require a percutaneous coronary intervention (PCI) to restore blood flow in narrowed or blocked coronary arteries.Q: Where is the stent typically visible during the procedure?
A: Thanks to its radiopaque properties, the stent is clearly visible under fluoroscopy, enabling clinicians to monitor and control its placement accurately during the intervention.Q: What safety features support infection control and product integrity?
A: Sterilized using either EO gas or Gamma irradiation, each stent is shipped in an individually sealed, sterile blister pack, which maintains sterility and reduces the risk of infection or contamination until the point of use.Q: How should the stent be stored prior to use?
A: Store the stent in a dry environment below 30C to maintain product integrity and sterility. This storage condition also helps preserve the drug stability and extends the stent's shelf life up to three years.Q: What advantages does the stent's flexible, low-recoil design provide?
A: The low-recoil design ensures that the stent maintains its expanded shape after deployment, supporting optimal scaffolding of the artery and consistent restoration of blood flow, while its flexibility helps accommodate varied vessel anatomies for improved clinical outcomes.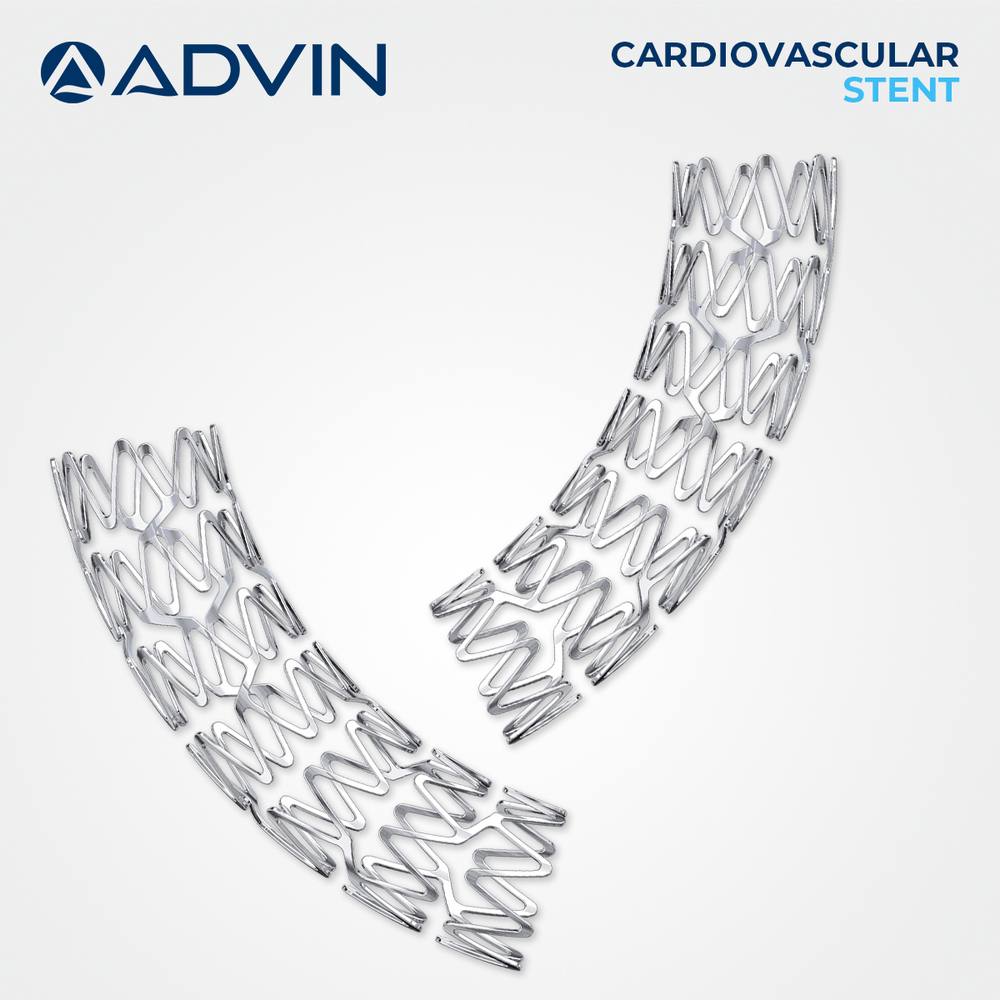






Price:
- 50
- 100
- 200
- 250
- 500
- 1000+

 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry



 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese