Coronary Artery Stents Cardiology
Price 200 USD ($)/ Piece
Coronary Artery Stents Cardiology Specification
- Storage Instructions
- Store in a cool, dry place away from direct sunlight
- Accuracy
- Optimal Vessel Placement
- Shelf Life
- 3 Years
- Function
- Restores Coronary Blood Flow
- Instruments Type
- Interventional Cardiology Device
- Usage Type
- Single Use
- Features
- Flexible, Biocompatible, Radiopaque, Drug Coating
- Measurement Range
- Diameter: 2.0-4.0 mm; Length: 8-38 mm
- Equipment Type
- Coronary Artery Stent
- Material
- Cobalt-Chromium Alloy
- Condition
- New
- Technology
- Drug Eluting
- Portable
- Yes
- Wall Mounted
- No
- Real-Time Operation
- Yes
- Noise Level
- Silent
- Operating Type
- Manual Delivery
- Use
- Cardiovascular Intervention / Angioplasty
- Power Source
- Not Required (Manual)
- Dimension (L*W*H)
- Varies by model (lengths: 8-38mm, diameter: 2.0-4.0mm)
- Weight
- Approximately 0.002-0.005 g
- Color
- Metallic Silver
- Sterilization Method
- Ethylene Oxide Sterile
- Surface Coating
- Polymer and Drug Layer (e.g., Sirolimus/Paclitaxel)
- Application
- Percutaneous Transluminal Coronary Angioplasty (PTCA)
- Packaging
- Sterile Blister Pack
- Compatibility
- Compatible with standard 0.014" guidewire
- Stent Type
- Balloon Expandable
- Radiopacity
- High for X-ray Visibility
- Delivery System
- Pre-mounted on Balloon Catheter
About Coronary Artery Stents Cardiology
Advin Health Care offers Coronary Artery Stents designed to meet international quality standards. These stents are widely used in cardiology for the treatment of narrowed or blocked coronary arteries, restoring optimal blood flow and supporting long-term vessel patency during percutaneous coronary intervention (PCI) procedures.
Key Features
- Advanced stent design for effective arterial support
- High radial strength for reliable vessel expansion
- Excellent flexibility and trackability in complex lesions
- Smooth and precise deployment system
- Biocompatible materials for enhanced safety and performance
Specifications
- Type: Coronary Artery Stent
- Material: Stainless Steel / Cobalt Chromium
- Variants: Drug Eluting Stent (DES) / Bare Metal Stent (BMS)
- Delivery System: Balloon-expandable system
- Sterilization: EO Sterilized
- Usage: Single-use
- Application: Treatment of coronary artery disease (CAD) in cardiology procedures
Certifications
CE, ISO 13485, and ISO 9001 certified
Product Keywords
Coronary Artery Stents, Cardiology Stents, Coronary Stent, PCI Stent, Drug Eluting Stent, Bare Metal Stent, Cardiac Stent
, , , , , ,
Stents de arteria coronaria, stents de cardiologa, stent coronario, stent para ICP, stent liberador de frmacos, stent metlico desnudo, stent cardaco
Stents coronaires, stents de cardiologie, stent coronaire, stent d'intervention coronarienne percutane (ICP), stent lution de mdicament, stent mtallique nu, stent cardiaque
Advanced Drug-Eluting Technology
These coronary artery stents utilize a sophisticated polymer coating infused with proven anti-restenotic drugs like Sirolimus or Paclitaxel. This modern approach significantly reduces the risk of vessel re-narrowing by gradually releasing the medication at the stent site, resulting in improved long-term vascular outcomes.
Precision Delivery and High Visibility
Each stent is balloon-expandable and pre-mounted on a balloon catheter, simplifying the deployment process. Their high radiopacity ensures clear visualization under X-ray, supporting optimal placement and reducing procedural risk. Cobalt-chromium construction offers a balance of strength and flexibility for diverse patient anatomies.
Safe, Sterile, and Reliable
Every stent is sterilized using reliable ethylene oxide, packaged in a secure sterile blister pack, and designed for single use to ensure patient safety. Compatibility with the standard 0.014" guidewire streamlines integration into established interventional cardiology procedures, making it convenient and dependable for healthcare professionals.
FAQ's of Coronary Artery Stents Cardiology:
Q: How is the coronary artery stent used during a PTCA procedure?
A: The stent, pre-mounted on a balloon catheter, is delivered via a standard 0.014" guidewire to the narrowed coronary segment. The balloon is then inflated, expanding the stent against the vessel wall to restore blood flow. The balloon is deflated and withdrawn, leaving the stent in place.Q: What benefits does the drug-eluting stent provide over bare metal options?
A: The polymer-drug layer, typically Sirolimus or Paclitaxel, minimizes the risk of restenosis by gradually releasing medication that inhibits tissue overgrowth, leading to sustained vessel patency compared to bare metal stents.Q: When should this type of stent be selected for a patient?
A: This stent is suitable for adult patients diagnosed with coronary artery blockages and indicated for percutaneous transluminal coronary angioplasty, especially where reducing the risk of restenosis is essential.Q: Where should the stents be stored before use?
A: Stents should be stored in a cool, dry place away from direct sunlight, ensuring the temperature and humidity remain stable to preserve the integrity and sterility of the device.Q: What is the process for ensuring the stent maintains sterility until use?
A: Each stent undergoes ethylene oxide sterilization and is sealed in a sterile blister pack. The packaging should not be opened until immediately prior to the procedure to maintain sterility.Q: Is the stent compatible with all guidewire sizes?
A: The stent is specifically designed for use with standard 0.014-inch guidewires, which are commonly used in interventional cardiology.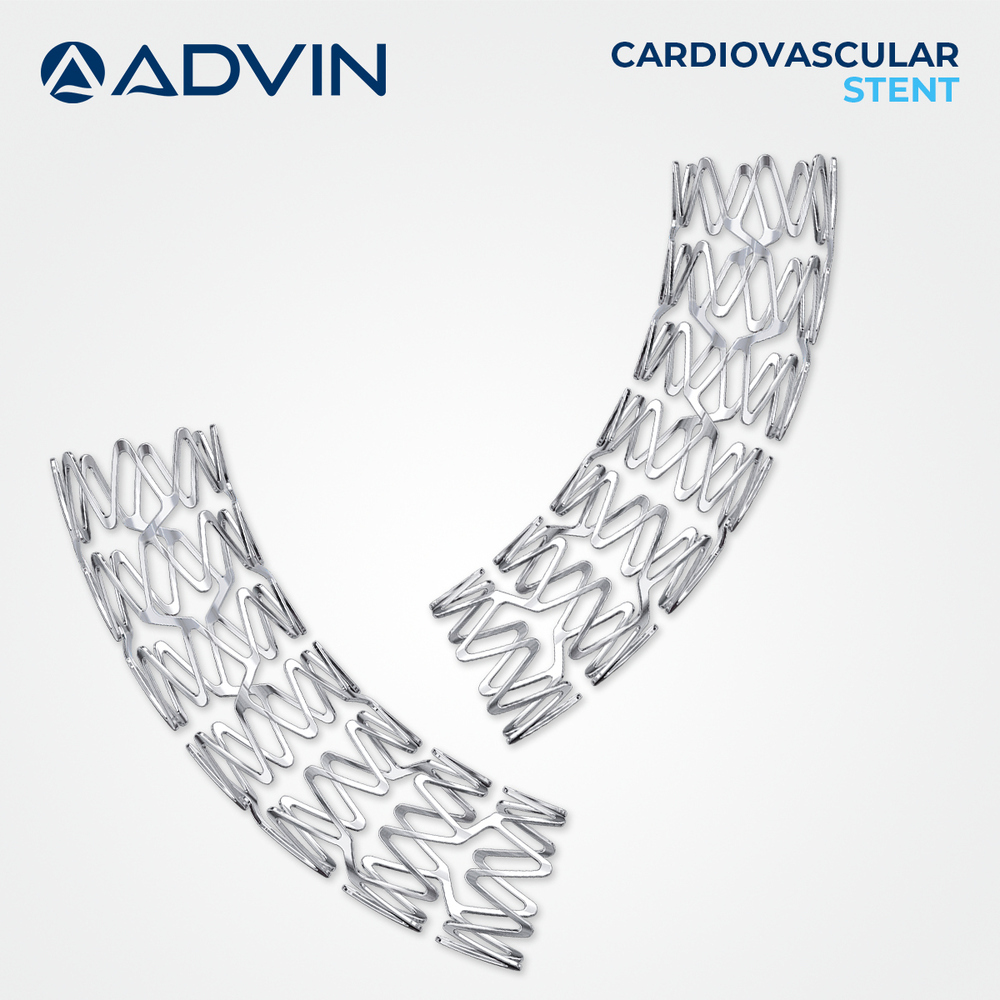




Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Cardiology Products Category
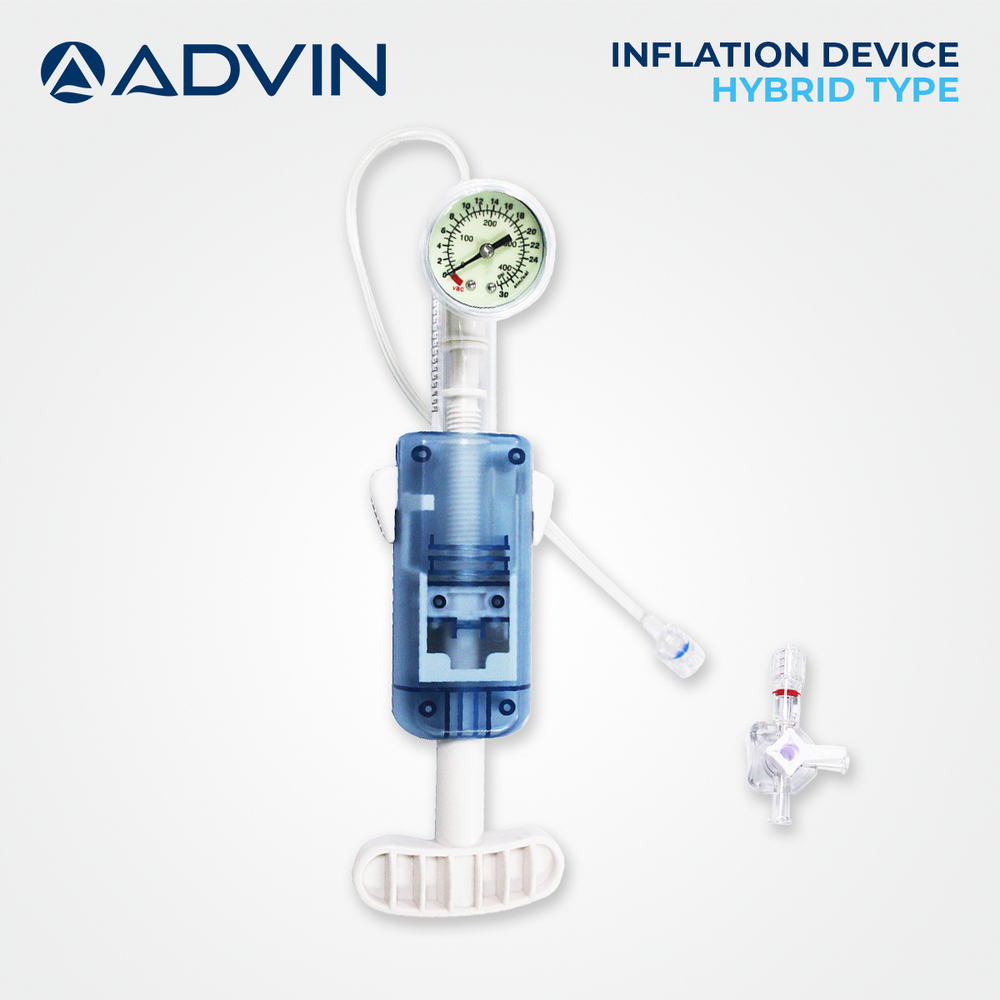
Interventional Trigger Inflator
Price 25 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Accuracy : 2% of Full Scale
Storage Instructions : Store in a cool, dry place away from sunlight
Color : Transparent body with blue plunger and dial
Wall Mounted : No
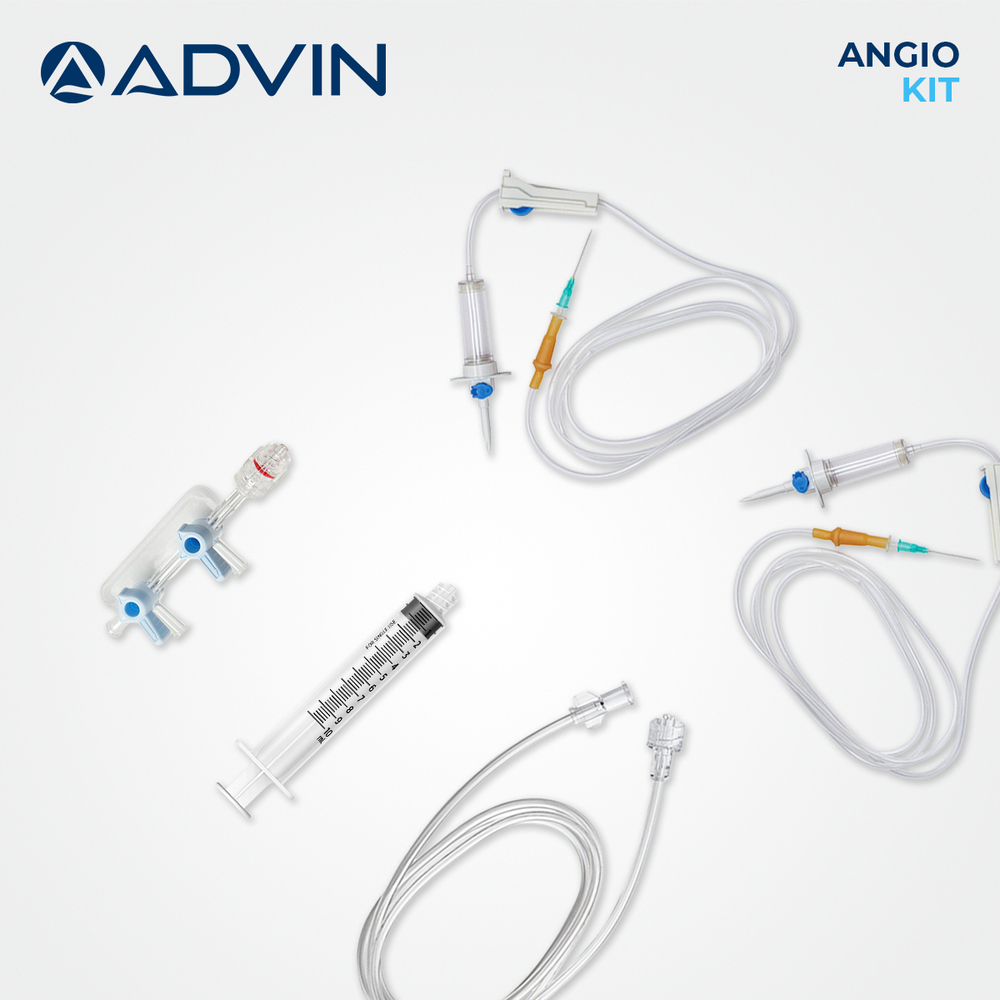
Cardiology Disposable Products
Price 25 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Accuracy : High Precision for Intended Medical Use
Storage Instructions : Store in a Cool, Dry Place, Away from Direct Sunlight
Color : Transparent, Blue, White, or as per Standard
Wall Mounted : No

 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry


 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese