Coronary Stent - Sirolium
Price 200 USD ($)/ Piece
Coronary Stent - Sirolium Specification
- Usage Type
- Single Use
- Function
- Revascularization of coronary arteries
- Accuracy
- High placement precision
- Features
- Biocompatible, Flexible, Non-corrosive
- Measurement Range
- Diameter 2.5 mm to 4.0 mm, Length 8 mm to 38 mm
- Instruments Type
- Interventional Cardiology
- Storage Instructions
- Store in a cool & dry place, avoid direct sunlight, keep in original packaging
- Shelf Life
- 3-5 Years
- Equipment Type
- Coronary Stent
- Material
- Cobalt Chromium Alloy
- Condition
- New
- Technology
- Drug Eluting Stent (DES)
- Portable
- Yes
- Wall Mounted
- No
- Real-Time Operation
- Yes
- Noise Level
- Silent
- Operating Type
- Manual deployment
- Use
- Coronary Artery Disease treatment
- Dimension (L*W*H)
- Vary by size (typical length: 8-38 mm, diameter: 2.5-4.0 mm)
- Weight
- Lightweight
- Color
- Metallic Silver
- Thrombogenicity
- Low
- Release Profile
- Sustained drug release up to 60 days
- Endothelialization
- Rapid, due to sirolimus coating
- Drug Coating
- Sirolimus
- Compatibility
- 5F, 6F, and 7F guiding catheters
- Sterilization
- ETO (Ethylene Oxide) sterilized
- Strut Thickness
- ~70-80 microns
- Radiopacity
- Enhanced for easy visualization
- Expansion Method
- Balloon expandable
- Delivery System
- Over-the-wire (OTW)
- Possible Balloon Diameter
- 2.5 - 4.0 mm
About Coronary Stent - Sirolium
Advin Health Care offers Coronary Stents Sirolimus Eluting designed to meet international quality standards. These drug-eluting stents (DES) are coated with sirolimus, an antiproliferative agent that helps reduce restenosis by inhibiting smooth muscle cell proliferation, ensuring long-term vessel patency and improved clinical outcomes in coronary artery disease treatment.
Key Features
- Sirolimus drug coating for reduced restenosis rates
- Controlled drug release for sustained therapeutic effect
- Thin strut design for enhanced flexibility and deliverability
- High radial strength for optimal vessel support
- Biocompatible polymer coating for safety and performance
Specifications
- Type: Drug Eluting Coronary Stent (Sirolimus)
- Material: Cobalt Chromium / Stainless Steel
- Drug: Sirolimus
- Coating: Biocompatible polymer-based drug coating
- Delivery System: Balloon-expandable system
- Sterilization: EO Sterilized
- Usage: Single-use
- Application: Treatment of coronary artery disease (CAD) via PCI procedures
Certifications
CE, ISO 13485, and ISO 9001 certified
Product Keywords
Sirolimus Eluting Stent, Drug Eluting Coronary Stent, DES Stent, Coronary Stent, Angioplasty Stent, Cardiac Stent
DES
, , , , ,
Stent liberador de sirolimus, stent coronario liberador de frmacos, stent DES, stent coronario, stent de angioplastia, stent cardaco
Stent lution de sirolimus, stent coronaire lution de mdicament, stent DES, stent coronaire, stent d'angioplastie, stent cardiaque
Optimal Drug Release Technology
Featuring sustained sirolimus release for up to 60 days, this stent inhibits excessive tissue growth, reducing the probability of restenosis. The advanced drug-coating technology ensures controlled drug delivery and accelerates the healing process within coronary arteries, supporting better patient outcomes and rapid vascular recovery.
Precision and Compatibility
Engineered for high placement accuracy, the stent offers compatibility with multiple guiding catheter sizes-5F, 6F, and 7F-allowing interventional cardiologists flexibility in diverse procedural settings. Its refined strut thickness and superior radiopacity facilitate precise positioning and clear visualization throughout deployment.
Reliable and Safe Performance
Manufactured from cobalt chromium alloy and sterilized with ETO, the stent is both non-corrosive and biocompatible. Its low-thrombogenicity profile, lightweight construction, and balloon-expandable deployment provide consistent safety and efficacy, making it suitable for various clinical scenarios.
FAQ's of Coronary Stent - Sirolium:
Q: How does the sirolimus coating benefit patients receiving this coronary stent?
A: The sirolimus coating delivers a sustained release of medication for up to 60 days, which suppresses excessive tissue regrowth and lowers the risk of restenosis, while also promoting rapid endothelial healing for improved patient recovery.Q: What is the deployment process for the Sirolium coronary stent?
A: The stent is manually deployed using a balloon-expandable over-the-wire (OTW) system. Compatible with 5F, 6F, and 7F guiding catheters, it is positioned within the coronary artery and expanded using an angioplasty balloon for precise placement.Q: When should this drug-eluting stent be used in coronary artery disease treatment?
A: This stent is suitable for revascularization in patients with narrowed or blocked coronary arteries, especially when long-term prevention of vessel re-narrowing and rapid healing are desired outcomes.Q: Where should the coronary stent be stored prior to use?
A: It should be stored in a cool, dry place, within its original packaging, and away from direct sunlight to maintain sterility and product integrity until use.Q: What is the benefit of the stent's enhanced radiopacity?
A: The enhanced radiopacity allows for easy and accurate visualization under fluoroscopy during procedures, ensuring precise placement and optimal clinical outcomes.Q: How does the stent ensure safety during use?
A: Made from biocompatible, non-corrosive cobalt chromium alloy and sterilized using ETO, the stent has a low potential for thrombogenicity, thus minimizing the risk of complications during and after implantation.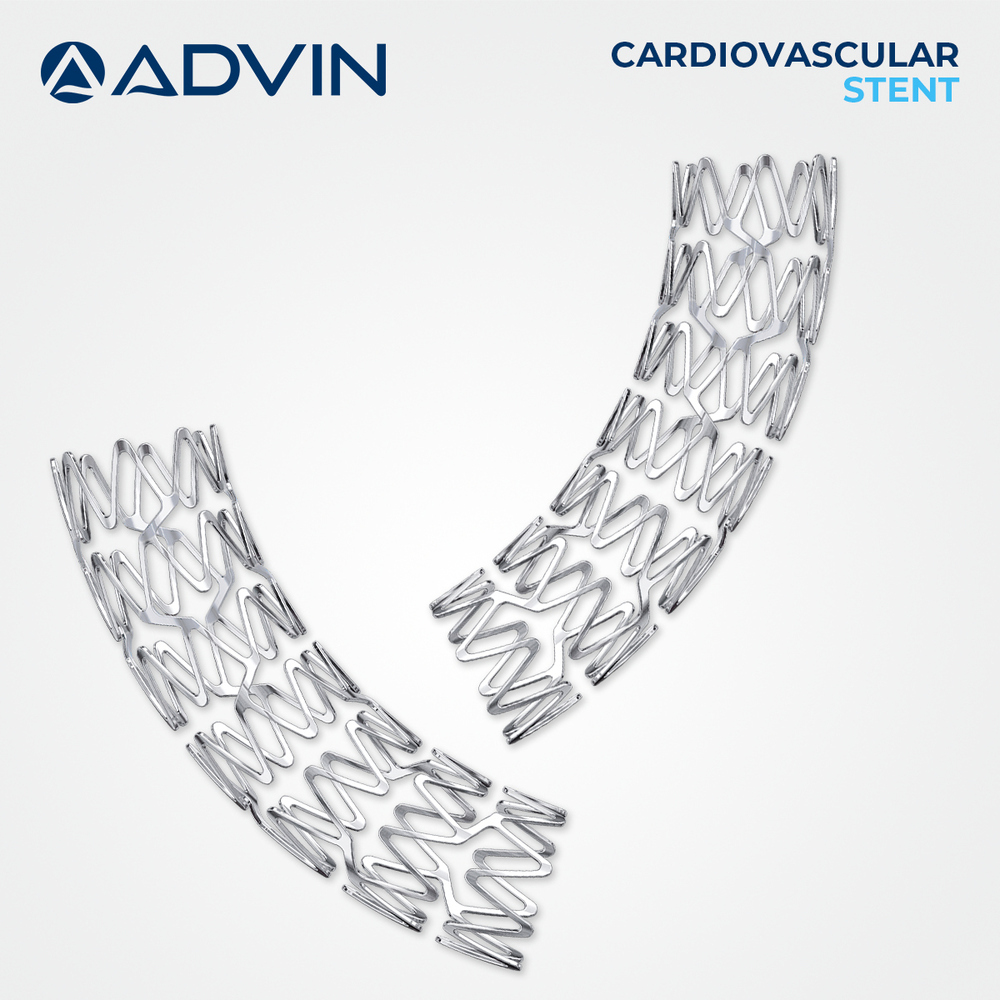




Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Cardiology Products Category
Cardiovascular Catheter
Price 25 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Technology : Catheterization Technology
Equipment Type : Cardiovascular Catheter
Weight : Lightweight (Typically <50g)
Condition : New
Advin Angioplasty Stent
Price 200 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Technology : Balloon Expandable
Equipment Type : Angioplasty Stent
Weight : Lightweight, < 10g
Condition : Other, Sterile, New

 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry


 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese