Craniotomy Drape
Price 5.62 USD ($)/ Piece
Craniotomy Drape Specification
- Function
- Provides a sterile barrier during craniotomy procedures
- Shelf Life
- 3-5 years (when stored properly)
- Features
- Sterile, Fluid-resistant, Tear-resistant, High barrier protection, Reinforced area for operating field, Adhesive drape area
- Instruments Type
- Surgical Drapes
- Usage Type
- Single-use
- Storage Instructions
- Store in a dry, cool place away from direct sunlight
- Equipment Type
- Craniotomy Drape
- Material
- Non-woven fabric
- Condition
- New
- Technology
- SMS (Spunbond-Meltblown-Spunbond) technology
- Portable
- Yes
- Wall Mounted
- No
- Real-Time Operation
- Yes
- Noise Level
- Silent
- Operating Type
- Disposable
- Use
- Craniotomy/Neurosurgery procedures
- Dimension (L*W*H)
- Approx. 250 cm x 300 cm (variable sizes available)
- Weight
- Lightweight
- Color
- Blue/Green
- Sterility
- Sterile EO (Ethylene Oxide) packed
- Application
- Ideal for use in neurosurgical, cranial and brain-related procedures
- Conformity
- Meets hospital/surgical standards
- Latex Content
- Latex-free
- Adhesive Zone
- Available with adhesive area for secure positioning
- Packing Type
- Individually packed in peelable pouch
- Fluid Collection Pouch
- Integrated fluid collection pouch included
About Craniotomy Drape
The Craniotomy Drape plays a vital role in maintaining aseptic conditions during complex brain and skull surgeries. Its design supports precise exposure of the operative area while helping manage fluids and maintain overall surgical cleanliness. The drape assists surgical teams by supporting organized workflow and consistent sterile coverage throughout the procedure. Nivda Drapes offers this disposable drape as a dependable solution that meets hospital standards for neurosurgical safety and hygiene.
Advanced Features
- Designed specifically for craniotomy and neurosurgical procedures
- Helps maintain a sterile and controlled surgical environment
- Supports fluid management during surgery
- Suitable for neurosurgical operation theaters and hospitals
غطاء جراØØ© الجمجمة، غطاء جراØØ© الأعصاب، غطاء جراØØ© الدماغ، غطاء جراØØ© الجمجمة المعقم، غطاء جراØÙŠ للاستخدام مرة واØدة، غطاء جراØØ© الأعصاب لغرÙØ© العمليات، غطاء جراØÙŠ غير منسوج
ХирургичеÑÐºÐ°Ñ Ð¿Ñ€Ð¾ÑÑ‚Ñ‹Ð½Ñ Ð´Ð»Ñ ÐºÑ€Ð°Ð½Ð¸Ð¾Ñ‚Ð¾Ð¼Ð¸Ð¸, нейрохирургичеÑÐºÐ°Ñ Ð¿Ñ€Ð¾ÑтынÑ, проÑÑ‚Ñ‹Ð½Ñ Ð´Ð»Ñ Ð½ÐµÐ¹Ñ€Ð¾Ñ…Ð¸Ñ€ÑƒÑ€Ð³Ð¸Ð¸, ÑÑ‚ÐµÑ€Ð¸Ð»ÑŒÐ½Ð°Ñ Ñ…Ð¸Ñ€ÑƒÑ€Ð³Ð¸Ñ‡ÐµÑÐºÐ°Ñ Ð¿Ñ€Ð¾ÑÑ‚Ñ‹Ð½Ñ Ð´Ð»Ñ ÐºÑ€Ð°Ð½Ð¸Ð¾Ñ‚Ð¾Ð¼Ð¸Ð¸, Ð¾Ð´Ð½Ð¾Ñ€Ð°Ð·Ð¾Ð²Ð°Ñ Ñ…Ð¸Ñ€ÑƒÑ€Ð³Ð¸Ñ‡ÐµÑÐºÐ°Ñ Ð¿Ñ€Ð¾ÑтынÑ, нейрохирургичеÑÐºÐ°Ñ Ð¿Ñ€Ð¾ÑÑ‚Ñ‹Ð½Ñ Ð´Ð»Ñ Ð¾Ð¿ÐµÑ€Ð°Ñ†Ð¸Ð¾Ð½Ð½Ð¾Ð¹, Ð½ÐµÑ‚ÐºÐ°Ð½Ð°Ñ Ñ…Ð¸Ñ€ÑƒÑ€Ð³Ð¸Ñ‡ÐµÑÐºÐ°Ñ Ð¿Ñ€Ð¾ÑтынÑ
Campo quirúrgico para craneotomÃa, campo quirúrgico para neurocirugÃa, campo quirúrgico para cirugÃa cerebral, campo quirúrgico estéril para craneotomÃa, campo quirúrgico desechable, campo quirúrgico para neurocirugÃa de quirófano, campo quirúrgico no tejido
Champ opératoire de craniotomie, champ opératoire de neurochirurgie, champ opératoire pour chirurgie cérébrale, champ opératoire stérile de craniotomie, champ opératoire jetable, champ opératoire de neurochirurgie, champ opératoire non tissé
Designed for Neurosurgery and Craniotomy
Specially crafted for use in neurosurgical and brain-related procedures, the Craniotomy Drape delivers optimal protection for the sterile field. Its reinforced operating area and secure adhesive zones support precise and safe surgical access, reducing the risk of contamination. The integrated fluid collection pouch efficiently manages fluids during complex surgical interventions.
Advanced Sterility and User Convenience
Packed using Ethylene Oxide (EO) sterilization, the drape maintains its sterility for a shelf life of up to 5 years when properly stored. Each drape is individually packaged in an easy-peel pouch, ensuring it remains uncontaminated prior to use. Its lightweight, portable design also facilitates effortless setup and disposal.
FAQ's of Craniotomy Drape:
Q: How is the Craniotomy Drape applied during surgical procedures?
A: The drape is placed over the operative field after patient preparation. The adhesive zone is gently pressed to the surrounding skin, ensuring it remains securely in position throughout the procedure, while the integrated pouch collects fluids effectively.Q: What materials are used in the manufacture of the Craniotomy Drape?
A: The drape is composed of SMS (Spunbond-Meltblown-Spunbond) technology non-woven fabric, offering high barrier protection, tear resistance, and fluid resistance. It is latex-free, reducing the risk of allergic reactions.Q: When should the Craniotomy Drape be used?
A: This drape is ideal for neurosurgical, cranial, and brain-related operations where a sterile barrier and effective fluid management are crucial for patient and staff safety.Q: Where can the Craniotomy Drape be implemented in the hospital setting?
A: It can be utilized in operating rooms equipped for neurosurgery or any setting requiring advanced infection control during cranial procedures.Q: What is the sterility process of the Craniotomy Drape?
A: The drape is sterilized using Ethylene Oxide (EO) gas, ensuring reliable sterility and long shelf life when stored in a cool, dry place away from sunlight.Q: Are there any specific storage requirements for maintaining the drape's sterility?
A: Yes, the drape should be stored in a dry, cool environment away from direct sunlight. Following these guidelines maintains the sterility and integrity until use.Q: What benefits does the integrated fluid collection pouch provide?
A: The built-in pouch enhances the procedure's safety and cleanliness by efficiently containing fluids, reducing the risk of contamination and simplifying postoperative cleanup.






Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Disposable Drapes Category
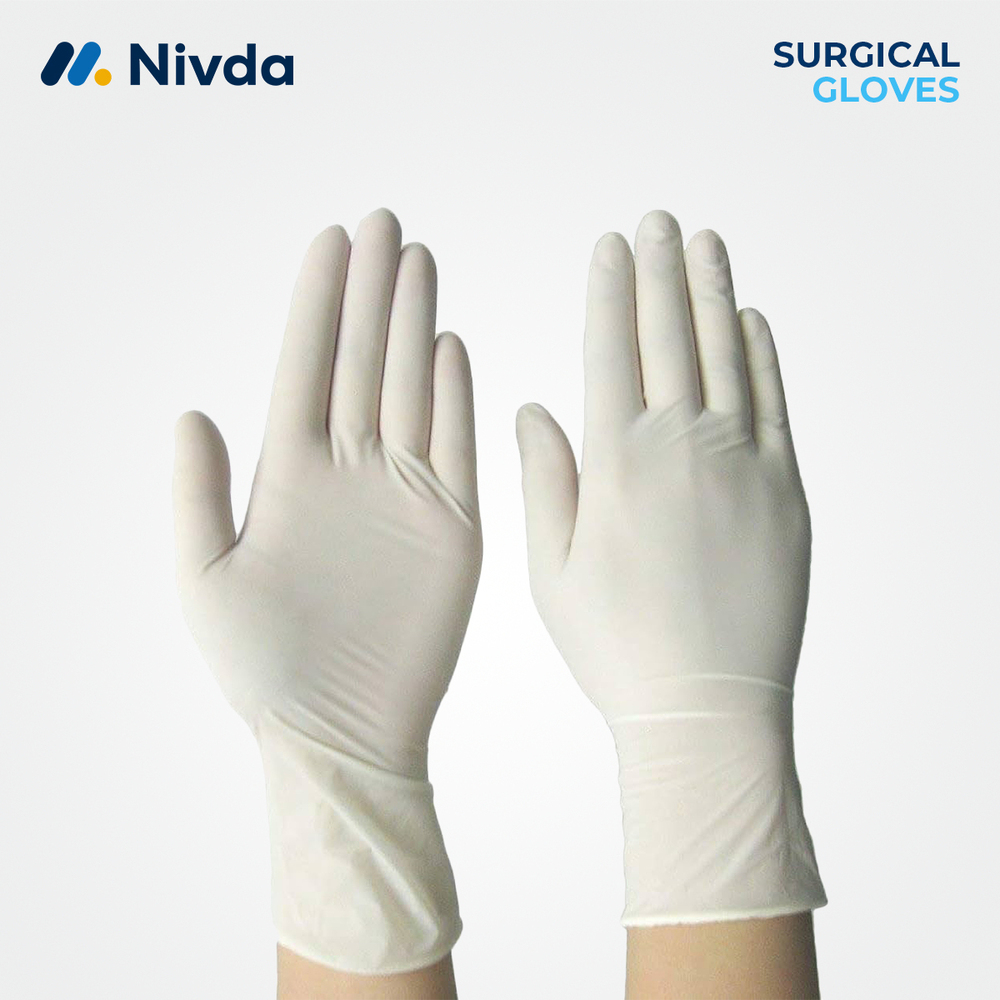
powder free latex surgical gloves 7
Price 2.15 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Storage Instructions : Store in a cool, dry place away from direct sunlight
Use : Surgical Procedures
Technology : PowderFree
Material : Latex
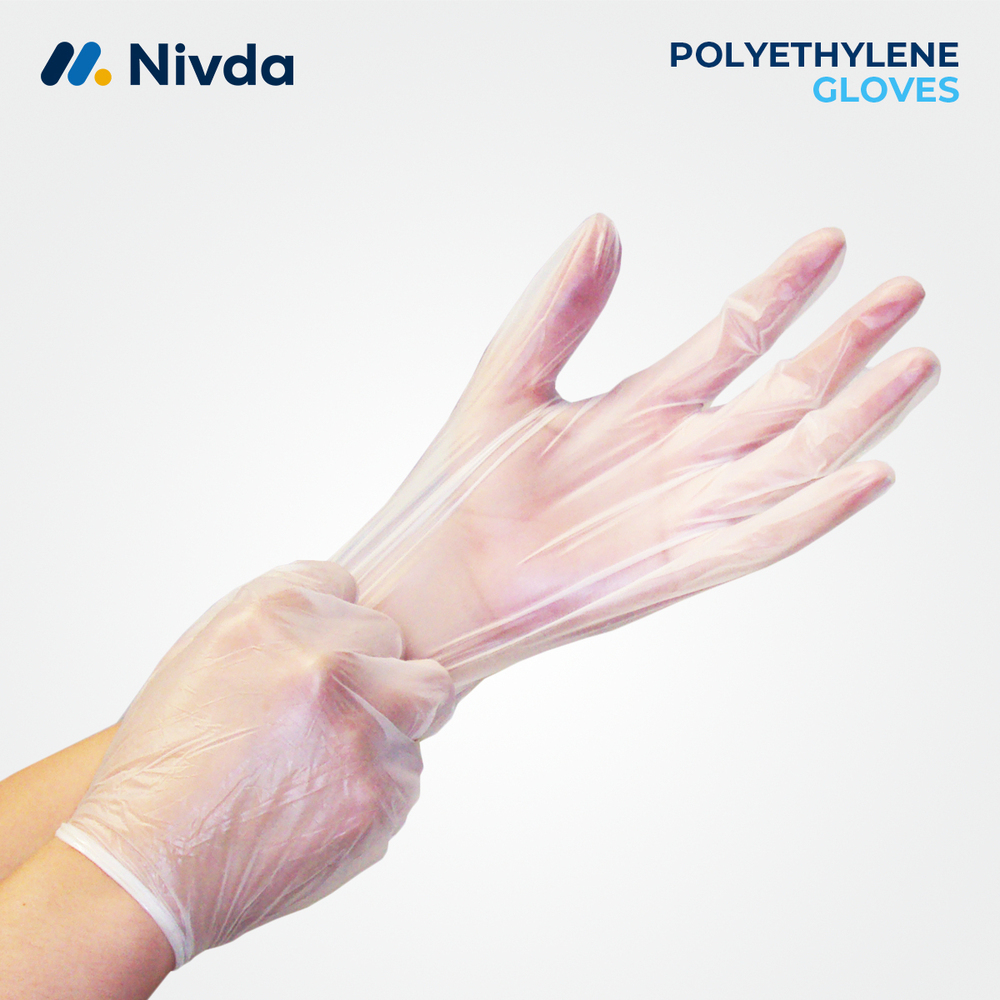
Polyethylene Gloves 50 GSM Large
Price 3.16 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Storage Instructions : Store in Cool, Dry Place, Away from Direct Sunlight
Use : Hand Protection
Technology : Blown Film Extrusion
Material : Other, Polyethylene (PE)
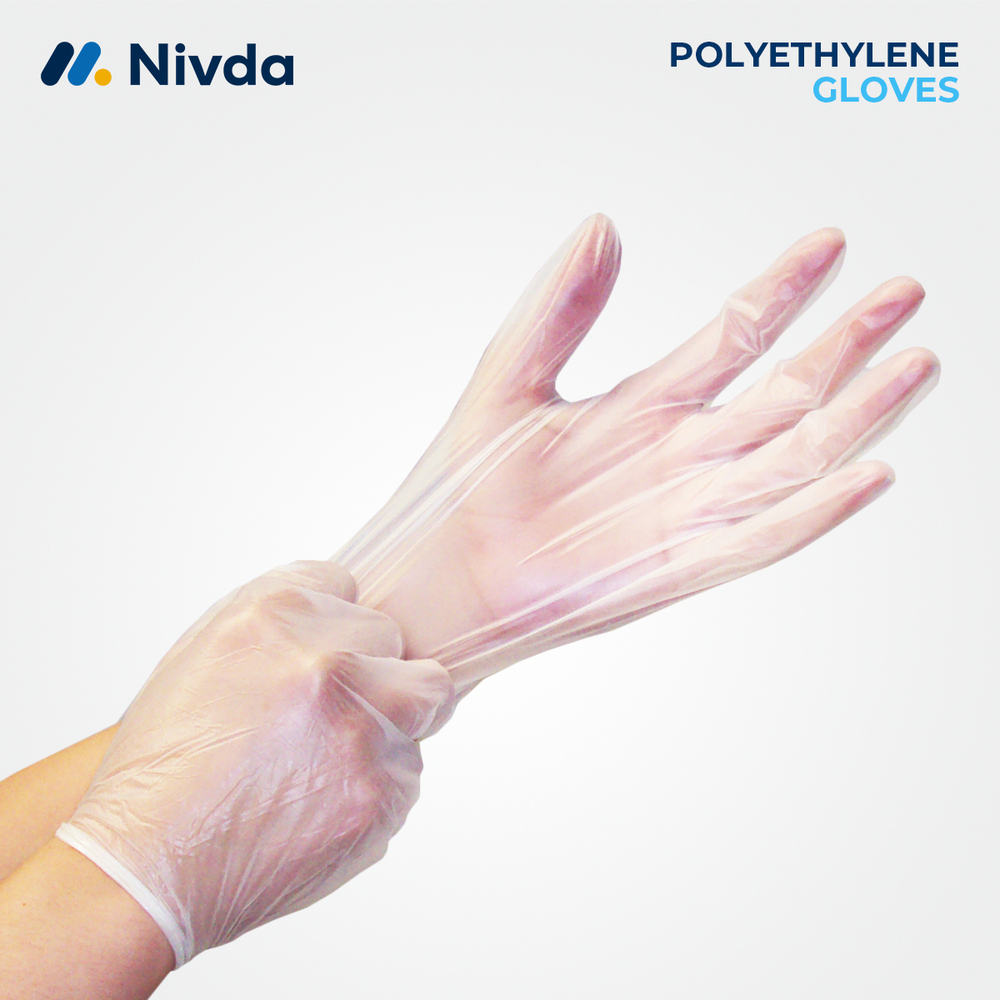
Polyethylene Gloves
Price 3.11 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Storage Instructions : Store in a cool, dry place away from direct sunlight
Use : Hand Protection
Technology : Cast Polyethylene Film
Material : Other, Polyethylene
Disposable Arthroscopy Drape Kit
Price 8.03 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Storage Instructions : Store in Cool, Dry Place; away from direct sunlight
Use : Surgical Procedures, Arthroscopy
Technology : Ultrasonic Sealing
Material : Other, SMS/SMMS Nonwoven Fabric

 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry

 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese