Disposable PTCA Kit
Price 18 USD ($)/ Piece
Disposable PTCA Kit Specification
- Usage Type
- Hospital, Clinical
- Function
- Assists in balloon angioplasty procedures
- Shelf Life
- 3-5 Years
- Instruments Type
- Interventional Cardiology Kit
- Accuracy
- Procedure-dependent
- Features
- Disposable, Ready-to-use, Sterile packed, All-in-one set
- Storage Instructions
- Store in dry, cool place away from direct sunlight
- Equipment Type
- Disposable PTCA Kit
- Material
- Medical Grade PVC, Polypropylene, Polyethylene
- Condition
- New
- Technology
- Sterile, Single Use
- Portable
- Yes
- Wall Mounted
- No
- Real-Time Operation
- Yes, for procedure support
- Operating Type
- Manual
- Use
- Percutaneous Transluminal Coronary Angioplasty (PTCA) procedures
- Dimension (L*W*H)
- Standard kit packaging size
- Weight
- Lightweight (Kit Form)
- Color
- Transparent/Blue (as per components)
- Expiry Marking
- Clearly mentioned on packaging
- Compatibility
- Compatible with major PTCA accessories
- Regulatory Compliance
- CE marked, ISO 13485 certified
- Latex Free
- Yes
- Packaging Type
- Individual blister pack
- Sterility
- Sterile, EO (Ethylene Oxide) processed
- Usability
- Single-use only
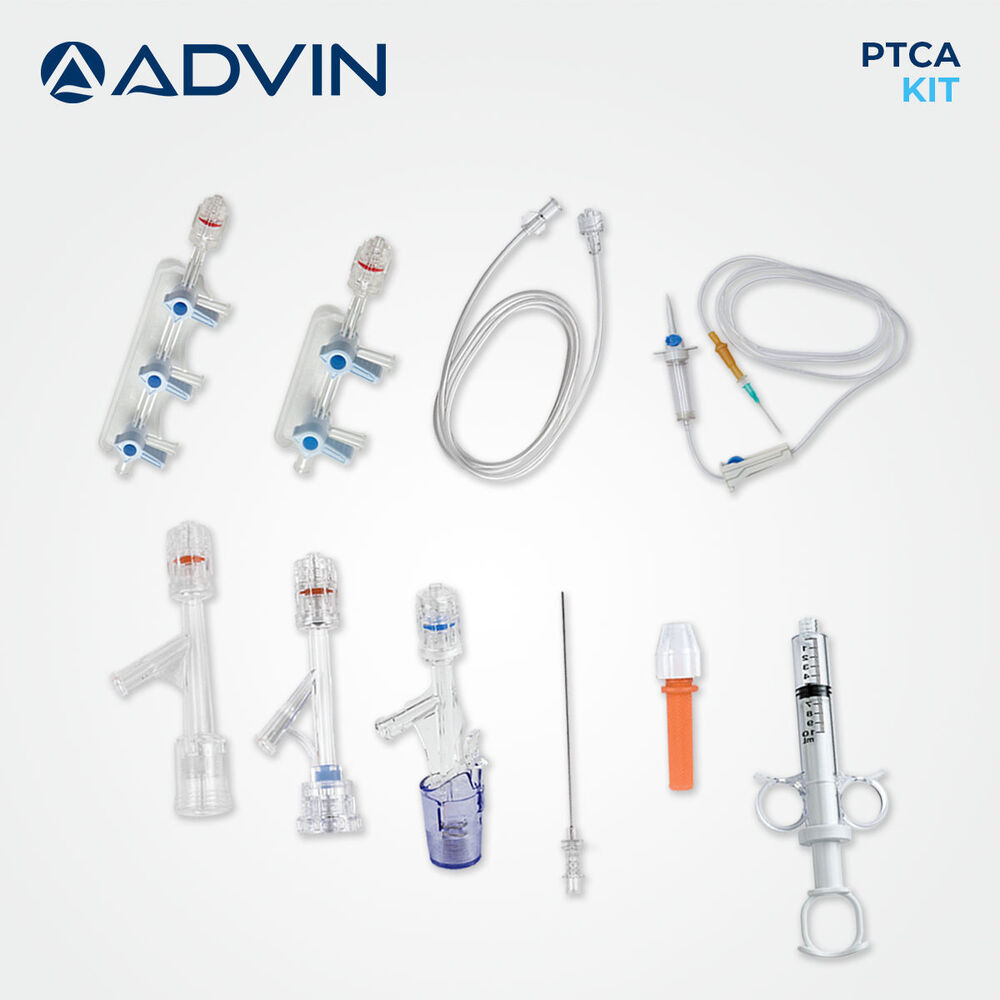
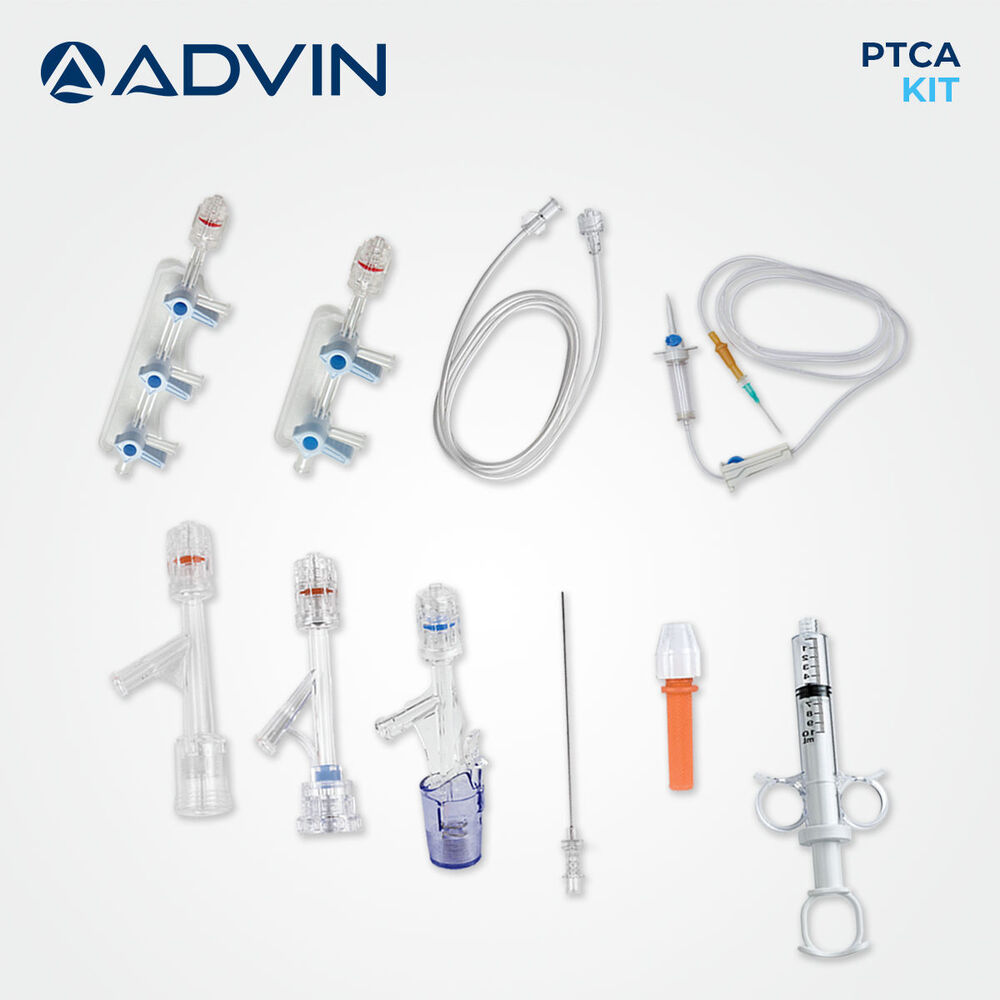
- Component List
- Includes PTCA guide wire, manifold, inflation device, syringes, hemostasis valve, torque device, drape, bowl, needles, and sterilized tray.
About Disposable PTCA Kit
The disposable PTCA kit is designed to support efficient coronary angioplasty by providing ready-to-use components for interventional procedures. Its single-use design helps maintain high hygiene standards and reduces the risk of cross-contamination. Advin Health Care focuses on delivering consistent quality and procedural reliability to meet clinical needs. The kit supports smooth workflow in cath labs and assists clinicians in performing coronary interventions with confidence.
Advanced FeaturesSingle-use design to support infection control Procedure-ready components for PTCA applications Designed for controlled and reliable coronary access Compatible with standard interventional cardiology devices
Certifications
We are certified withA CE, ISO 13485, and ISO 9001A standards.
Product Keywords
Disposable PTCA Kit, Single-Use PTCA Kit, PTCA Kit, Coronary Angioplasty Kit, Interventional Cardiology Kit, Angioplasty Procedure Kit
UUU1 UU1U UaU 1U UU UU UU3a U U U UUU1 UU1U UaU 1U UU UU UU3a U U U UUU1 UU1U UaU 1U UU UU UUU1 UU1U UaU UUU1 UUU Ua UU UUU1 a UU1U
1234N342N1 1234N N , 1234N N 34 1234N34234334 N34N34212 N, 1234N N , 1234N N o34N3412N12341 123 34NN o , 1234N N 12NN212N 341212341 oN 34343 , 1234N N N34N NNN 123 34NN o
Kit desechable para PTCA, kit de PTCA de un solo uso, kit de PTCA, kit de angioplastia coronaria, kit de cardiologAa intervencionista, kit para procedimiento de angioplastia
Kit d'angioplastie coronaire transluminale percutanAe (ACTP) A usage unique, kit d'angioplastie coronaire, kit de cardiologie interventionnelle, kit pour procAdure d'angioplastie
Comprehensive, Ready-to-Use Solution
This PTCA kit provides all critical components required for balloon angioplasty procedures in one sterile, disposable package. Eliminating the need for pre-procedure assembly, it streamlines workflow, reduces preparation time, and ensures every item is available exactly when needed. The kit's compatibility with leading PTCA accessories further enhances procedural efficiency and outcome reliability.
Strict Sterility and Quality Compliance
Processed with Ethylene Oxide (EO) for effective sterilization, each kit meets rigorous CE marking and ISO 13485 certification standards. The kit is latex-free and supplied in a protective blister pack, reducing contamination risk and maximizing patient safety. Clear expiry markings and a 3-5 year shelf life provide additional quality assurance for healthcare providers.
Convenient and Portable for Clinical Use
Lightweight and compact, the Disposable PTCA Kit is easily stored and transported within hospital and clinic environments. Its all-in-one, single-use design minimizes the risk of infection, supports rapid case turnover, and is particularly suitable for interventional cardiology teams seeking dependable, on-demand procedural support.
FAQ's of Disposable PTCA Kit:
Q: How do I use the Disposable PTCA Kit during angioplasty procedures?
A: Simply unwrap the sterile blister pack in an aseptic environment and utilize each component-such as guide wire, inflation device, and syringes-according to the standard PTCA procedure protocol. The all-in-one packaging ensures every essential tool is on hand and ready for immediate use.Q: What items are included in this Disposable PTCA Kit?
A: The kit contains a PTCA guide wire, manifold, inflation device, syringes, hemostasis valve, torque device, drape, bowl, needles, and a sterilized tray-all fabricated from premium medical-grade materials.Q: When should I use this kit in a clinical setting?
A: The Disposable PTCA Kit is designed for use during Percutaneous Transluminal Coronary Angioplasty (PTCA) procedures in hospital cath labs or interventional cardiology departments, providing essential support on a single-use basis.Q: Where is the best place to store the PTCA Kit?
A: For optimal shelf life and sterility, the kit should be kept in a dry, cool area away from direct sunlight, as recommended on the packaging.Q: What are the key benefits of using a single-use, disposable PTCA Kit?
A: Using a disposable kit minimizes infection risks, ensures sterility for each patient, saves preparation time, and prevents cross-contamination. The all-in-one design also reduces inventory management complexity.Q: Is the PTCA Kit compatible with accessories from other brands?
A: Yes, the kit is designed for broad compatibility with major PTCA accessories available in most medical facilities, facilitating seamless integration into standard workflows.Q: How can I confirm the kit's sterility and expiry?
A: Each kit undergoes thorough EO sterilization, contains clear expiry markings on the packaging, and meets CE and ISO 13485 certification requirements, ensuring ongoing sterility and reliability until the marked expiration date.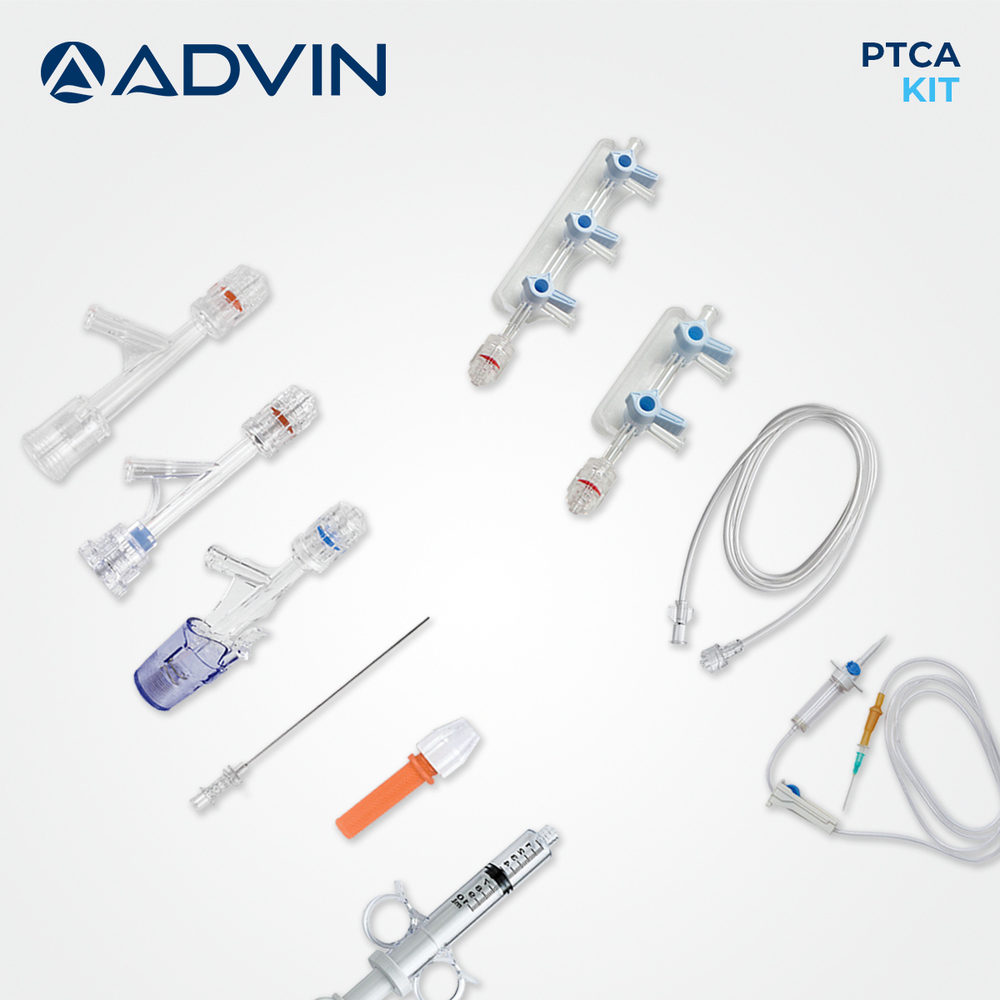




Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Cardiology Products Category
Flexible Guiding Catheter
Price 40 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Technology : Advanced extrusion, Radiopaque tip
Equipment Type : Flexible Guiding Catheter
Function : Guidance and support for interventional devices
Usage Type : Single Use (Disposable)
Cardiovascular Femoral Sheath
Price 7.00 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Technology : Advanced Sheath Design
Equipment Type : Cardiovascular Femoral Sheath
Function : Facilitates Introduction of Catheters and Wires
Usage Type : Single Use
Endovascular Capture Basket
Price 70 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Technology : Endovascular retrieval and capture
Equipment Type : Endovascular Capture Basket
Function : Vascular foreign body retrieval
Usage Type : Singleuse, disposable

 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry



 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese