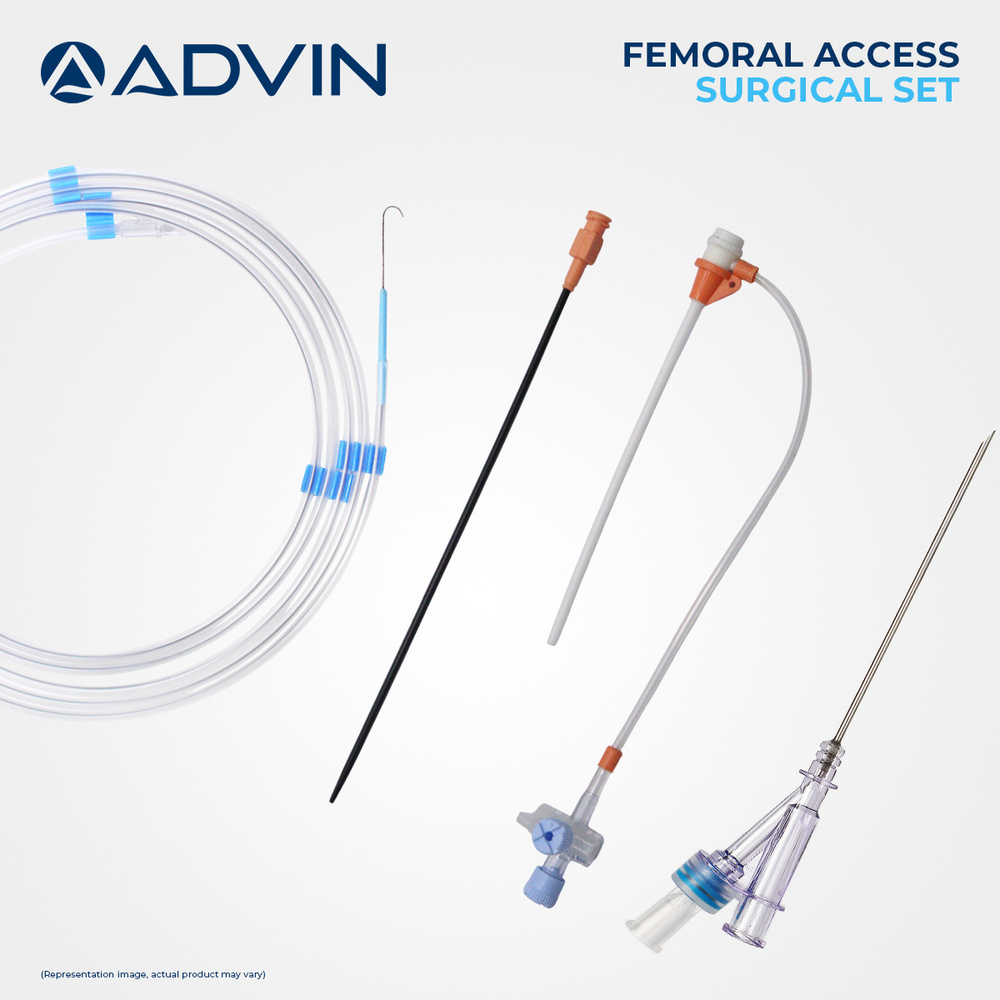
Disposable Trans Radial Sheath
Price 6 USD ($)/ Piece
Disposable Trans Radial Sheath Specification
- Shelf Life
- 3 Years
- Accuracy
- High Precision Access
- Measurement Range
- 4F, 5F, 6F, 7F sizes
- Instruments Type
- Interventional Cardiology Consumable
- Features
- Kink-resistant, Flexible Tip, Smooth Insertion, Easy-to-see Markings, Hydrophilic Coated
- Function
- Introducer Sheath for Radial Access
- Storage Instructions
- Store in Cool, Dry Place; Avoid Direct Sunlight
- Usage Type
- Disposable
- Equipment Type
- Disposable Trans Radial Sheath
- Material
- Other
- Technology
- Advanced Hydrophilic Coating
- Portable
- Yes
- Wall Mounted
- No
- Real-Time Operation
- Yes
- Noise Level
- Silent Operation
- Operating Type
- Manual
- Use
- Radial Artery Access
- Dimension (L*W*H)
- Varies by French Size (e.g., 10 cm length)
- Weight
- 2-10 g (depends on size)
- Color
- Transparent with Color-coded Hub
- Guidewire Compatibility
- 0.021'' - 0.038''
- Sterilization Method
- EO (Ethylene Oxide) Gas
- Tip Design
- Atraumatic, Tapered
- Latex Content
- Latex Free
- Sheath Sizes
- 4F, 5F, 6F, 7F (French) Available
- Intended Use
- Cardiac and Peripheral Vascular Procedures
- Connector
- Luer Lock
- Puncture Needle Included
- Yes (Standard in each pack)
- Hub Type
- Ergonomically Designed, Color-coded
- Packaging
- Individual Blister Pack
- Coating Type
- Hydrophilic surface for enhanced patient comfort and easy insertion
- Compliance
- ISO 13485, CE Marked
About Disposable Trans Radial Sheath
Advin Health Care is a trusted manufacturer of Disposable Trans Radial Sheaths designed to meet international quality standards. These sheaths are used in interventional cardiology procedures to provide safe and efficient vascular access through the radial artery while ensuring sterility and minimizing the risk of cross-contamination.
Key Features
- Single-use design to maintain sterility and prevent cross-contamination
- Smooth and atraumatic insertion for radial artery access
- Hydrophilic coating for reduced friction and enhanced patient comfort
- Flexible and kink-resistant construction for reliable performance
Certifications
We are certified with CE, ISO 13485, and ISO 9001 standards.
Product Keywords
Disposable Trans Radial Sheath, Radial Artery Sheath, Single-Use Sheath, Interventional Cardiology Sheath, Vascular Access Device
oU U UU UU1 U UU3a U U U oU U UU UU1 U oU UU3a U UU U oU UUU Ua UU U2 UUUU UU1U
1234N342N1 NN12NN N12N1 oNNN, oNNN N NN2341 NNN , oNNN N 34 1234oNN1234334 N34N34212 N, oNNN N 12NN212N 341212341 oN 34343 , NNNN341NN234 N N34NN NN34334 34NNN.
Vaina transradial desechable, vaina para arteria radial, vaina de un solo uso, vaina para cardiologAa intervencionista, dispositivo de acceso vascular
Gaine radiale jetable, gaine pour artA re radiale, gaine A usage unique, gaine de cardiologie interventionnelle, dispositif d'accA s vasculaire
Seamless Radial Access
Engineered for smooth, comfortable arterial entry, the Disposable Trans Radial Sheath uses advanced hydrophilic technology. This design significantly minimizes resistance during insertion, promoting patient safety and reducing procedure time. Its color-coded ergonomic hub and kink-resistant, flexible tip further optimize handling, making it an excellent choice for interventional cardiology specialists.
Safety and Compliance Assured
Every sheath is manufactured from medical-grade polyurethane, individually sterilized using EO (Ethylene Oxide) gas, and packaged in a secure blister pack. Fully latex-free and adhering to ISO 13485 standards with CE marking, the product guarantees the highest levels of patient safety and regulatory compliance for global hospital and clinical use.
Versatile, Precise, and User-Friendly
The sheath is compatible with guidewires ranging from 0.021 to 0.038 inches, supporting a broad array of diagnostic and interventional procedures. The tapered, atraumatic tip, easy-to-see markings, and luer lock connector contribute to high accuracy and secure operations. Its lightweight, portable, and silent operation design ensures comfort for both operator and patient.
FAQ's of Disposable Trans Radial Sheath:
Q: How is the Disposable Trans Radial Sheath used during cardiac or peripheral vascular procedures?
A: The sheath serves as an introducer for catheters and guidewires, facilitating safe and precise radial artery access. After puncturing the artery with the included needle and guidewire, the sheath is advanced into position, providing a smooth conduit for further interventional instruments.Q: What sizes are available and how do I select the right one?
A: This sheath is available in 4F, 5F, 6F, and 7F (French) sizes. The appropriate size depends on the specific procedure and instruments used, as well as patient anatomy. Medical professionals should select the size that best fits procedural and patient requirements.Q: Where should the Disposable Trans Radial Sheath be stored before use?
A: Store the sheath in a cool, dry area away from direct sunlight. Proper storage conditions ensure the integrity and sterility of the device until the expiration date (3-year shelf life).Q: What benefits does the hydrophilic coating provide during insertion?
A: The advanced hydrophilic surface significantly reduces friction, enabling smoother, less traumatic sheath advancement. This design lowers discomfort for the patient and can minimize the risk of arterial injury or spasm.Q: What is the process for ensuring the sheath remains sterile prior to use?
A: Each sheath is sterilized by EO (Ethylene Oxide) gas and packaged in an individual blister pack. Always check the packaging for damage or signs of compromise before opening, and use the sheath immediately after opening to maintain sterility.Q: When should the Disposable Trans Radial Sheath be discarded?
A: The sheath is intended for single use only. After completion of the procedure, it must be safely disposed of according to standard biohazard protocols to prevent cross-contamination or infection.Q: What are the main user benefits for clinicians using this sheath?
A: Clinicians benefit from its high kink resistance, flexible and atraumatic tapered tip, ergonomic hub, clear size coding, and precise luer lock connector. These features enhance handling, control, and safety throughout interventional procedures.






Price:
- 50
- 100
- 200
- 250
- 500
- 1000+

 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry


 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese