Epidural Injection Kit
Price 8.5 USD ($)/ Piece
Epidural Injection Kit Specification
- Instruments Type
- Anesthesia procedure kit
- Measurement Range
- Graduated markings on syringe (up to 20 ml)
- Frequency
- Single-use
- Accuracy
- High precision for drug administration
- Shelf Life
- 5 years (unopened)
- Function
- To provide safe and effective epidural injections
- Storage Instructions
- Store in a cool, dry place away from sunlight
- Usage Type
- Hospital and clinical
- Features
- Ergonomic design, graduated markings, Luer lock compatibility, sterile packaging
- Equipment Type
- Epidural Injection Kit
- Material
- Medical grade plastic and stainless steel
- Condition
- New
- Technology
- Sterile, single-use disposable technology
- Portable
- Yes
- Wall Mounted
- No
- Real-Time Operation
- Yes
- Noise Level
- Silent operation
- Operating Type
- Manual
- Use
- For administering epidural anesthesia
- Power Source
- Not required (manual)
- Dimension (L*W*H)
- Standard kit size (varies per component), typically around 25 x 15 x 5 cm
- Weight
- Approx. 200 grams (total kit)
- Color
- Transparent/clear and blue components
- Catheter Length
- Typically 80-100 cm (flexible, kink-resistant)
- Latex Free
- Yes (latex-free components)
- Manufacturer Warranty
- Provided as per manufacturer policy
- Packaging
- Individually packed in sterile, peel-open blister trays
- Regulatory Compliance
- ISO 13485 certified and CE marked
- Needle Gauge
- 16G or 18G (depending on kit variant)
- Sterility
- Gamma sterilized and pyrogen-free
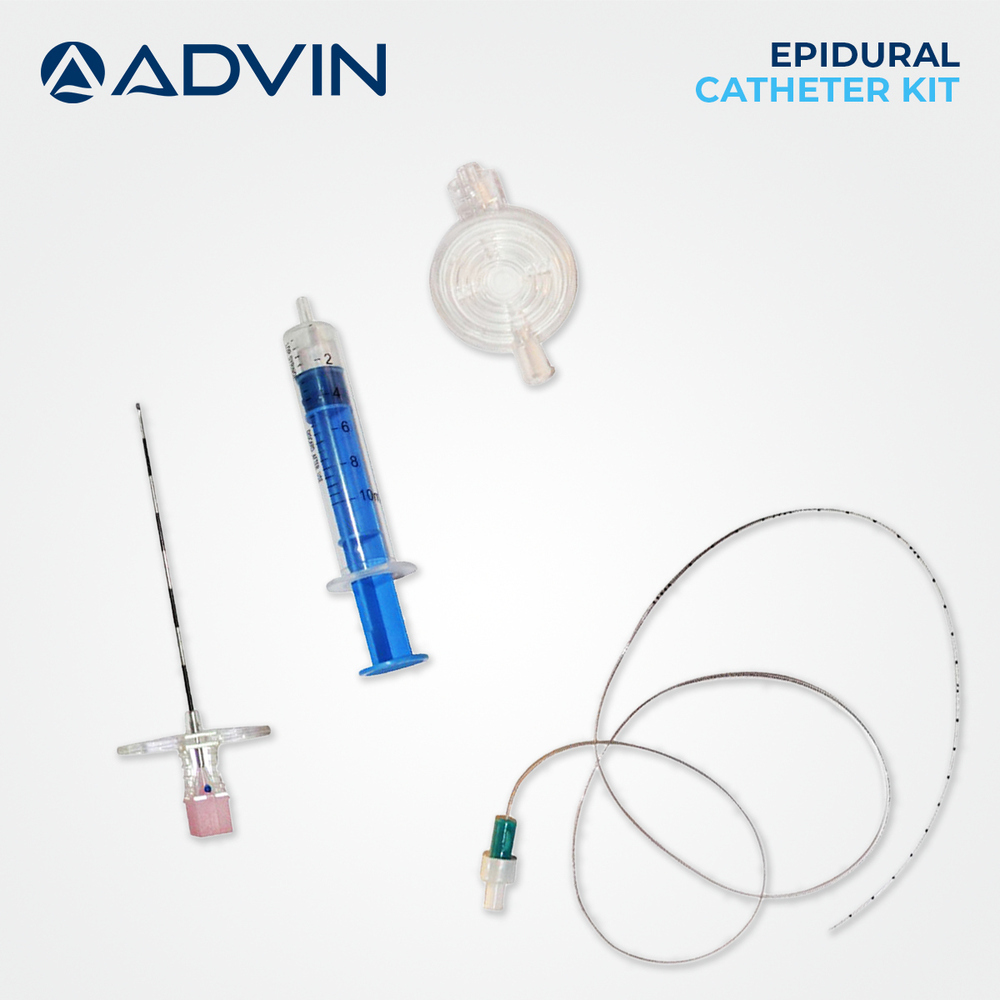
- Kit Components
- Epidural needle (Tuohy), LOR syringe, catheter, filter, drape, connector, specimen label, dressing, and adhesive plasters
About Epidural Injection Kit
Advin Health Care is a trusted manufacturer of Epidural Injection Kits designed to meet international quality standards. These kits are developed for precise and controlled administration of medications into the epidural space, supporting effective pain management in surgical, obstetric, and chronic pain procedures.
Key Features
- Designed for accurate epidural drug delivery
- Precision Tuohy needle for controlled epidural access
- Smooth and flexible catheter for consistent infusion
- High-quality components ensuring reliability and safety
- Sterile, single-use design to minimize infection risk
Specifications
- Type: Epidural Injection Kit
- Needle Type: Tuohy Needle
- Catheter Material: Medical-grade flexible polymer
- Sterilization: EO Sterilized
- Usage: Single-use
- Application: Epidural anesthesia, pain management, and therapeutic injections
Certifications
CE, ISO 13485, and ISO 9001 certified
Product Keywords
Epidural Injection Kit, Epidural Anesthesia Kit, Pain Injection Kit, Epidural Set, Anesthesia Injection Kit
, , , , .
Kit de inyeccin epidural, kit de anestesia epidural, kit de inyeccin para el dolor, set epidural, kit de inyeccin de anestesia
Kit d'injection pridurale, kit d'anesthsie pridurale, kit d'injection pour la douleur, kit pridural, kit d'injection d'anesthsie
Comprehensive Components for Safe Anesthesia
This epidural injection kit includes every necessary element for seamless administration of epidural anesthesia. The kit's design ensures reliability with graduated markings for precise drug dosage, a kink-resistant catheter for smooth delivery, and an ergonomic layout that supports clinical safety and efficiency throughout the procedure.
Stringent Quality and Sterility Standards
Gamma sterilized and entirely latex-free, the kit eliminates allergen risks and maintains sterility until use. Each set is certified under ISO 13485 and bears the CE mark, underscoring adherence to international quality and safety benchmarks for medical devices in hospital and clinic applications.
Flexible and Convenient Usage
The kit's components are lightweight, portable, and disposably designed for single-use. It is manually operated, requires no external power source, and is packaged for quick, peel-open access in demanding operating room situations, offering maximum convenience to healthcare providers.
FAQ's of Epidural Injection Kit:
Q: How is the Epidural Injection Kit used during an anesthesia procedure?
A: The kit is used by medical professionals for epidural administrations. The Tuohy needle is inserted into the epidural space using the LOR syringe, after which the catheter is threaded through the needle for continuous or single-shot anesthesia delivery. All steps are performed using the provided sterile, single-use components.Q: What components are included in the epidural injection kit?
A: Each kit contains a Tuohy epidural needle (16G or 18G), loss-of-resistance syringe (up to 20 ml), flexible catheter (80-100 cm), filter, drape, connector, specimen label, dressing, and adhesive plasters, conveniently packaged for one-time, sterile use.Q: When should the kit be used, and who is it intended for?
A: This kit is designed for use during hospital and clinical procedures requiring epidural anesthesia. It is intended for trained anesthesia practitioners and is to be used only when indicated for patient care.Q: Where should the Epidural Injection Kit be stored prior to use?
A: Store the unopened kit in a cool, dry place away from direct sunlight to preserve its sterility and optimal performance throughout its five-year shelf life.Q: What are the safety and sterility measures maintained with this kit?
A: All components are gamma sterilized, pyrogen-free, and latex-free. The kit conforms to ISO 13485 and CE standards, and comes individually packed to prevent contamination, safeguarding both patient and provider.Q: How does the kit benefit healthcare providers and patients?
A: It streamlines the preparation for epidural procedures by offering all required components in one sterile, ready-to-use package, reducing the risk of infection and ensuring precise drug administration through its graduated and ergonomic components.Q: What is the process for disposing of the kit after use?
A: Following the procedure, all used kit components should be disposed of as per hospital or clinical guidelines for single-use medical devices, typically in designated sharps and biohazard containers.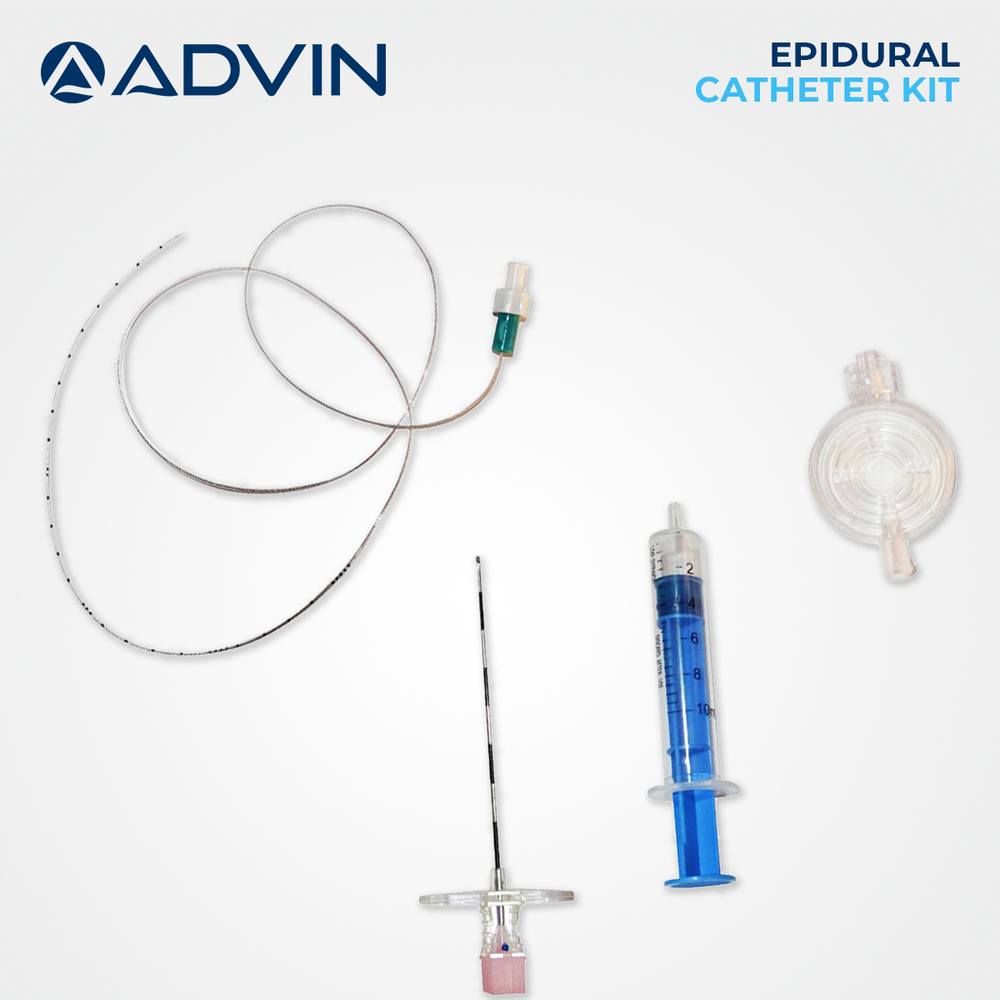



Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Cardiology Products Category
Cardiovascular Sheath
Price 8 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Real-Time Operation : Yes
Wall Mounted : No
Weight : Approx. 30 gm
Portable : Yes
Angiography 2-Way Manifold
Price 2 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Real-Time Operation : Yes
Wall Mounted : No
Weight : Lightweight (approx. 2030g)
Portable : Yes

 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry




 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese