Infection Prevention in Surgery
Price 9.60 USD ($)/ Piece
Infection Prevention in Surgery Specification
- Features
- Comprehensive infection prevention, sterile packaging, easy handling, single-use items
- Usage Type
- Hospital/Clinic surgery rooms
- Frequency
- Single use
- Instruments Type
- Surgical barrier kit
- Shelf Life
- 2 years
- Storage Instructions
- Store in a cool, dry place away from direct sunlight
- Function
- Prevention of infection and maintenance of sterile field
- Equipment Type
- Infection Prevention in Surgery Kit
- Material
- Medical-grade components (e.g., sterile drapes, gloves, masks, disinfectants)
- Condition
- New
- Technology
- Barrier technology and antimicrobial filtration
- Portable
- Yes
- Wall Mounted
- No
- Real-Time Operation
- Yes
- Noise Level
- Silent
- Operating Type
- Manual
- Use
- Sterile field preparation and infection control in surgical settings
- Dimension (L*W*H)
- Standard kit size, 35 cm x 22 cm x 8 cm (approx.)
- Weight
- 1.2 kg (approx.)
- Color
- Blue/White (as per standard medical supplies)
- Packaging Type
- Sterile, tamper-proof packaging
- Latex Content
- Latex-free
- Sterilization Method
- Ethylene oxide sterilized
- Compliance Standard
- CE/ISO 13485 certified
- Target Users
- Surgeons, surgical staff, and operating room personnel
- Application
- Preoperative and intraoperative infection control
- Disposability
- Single use only
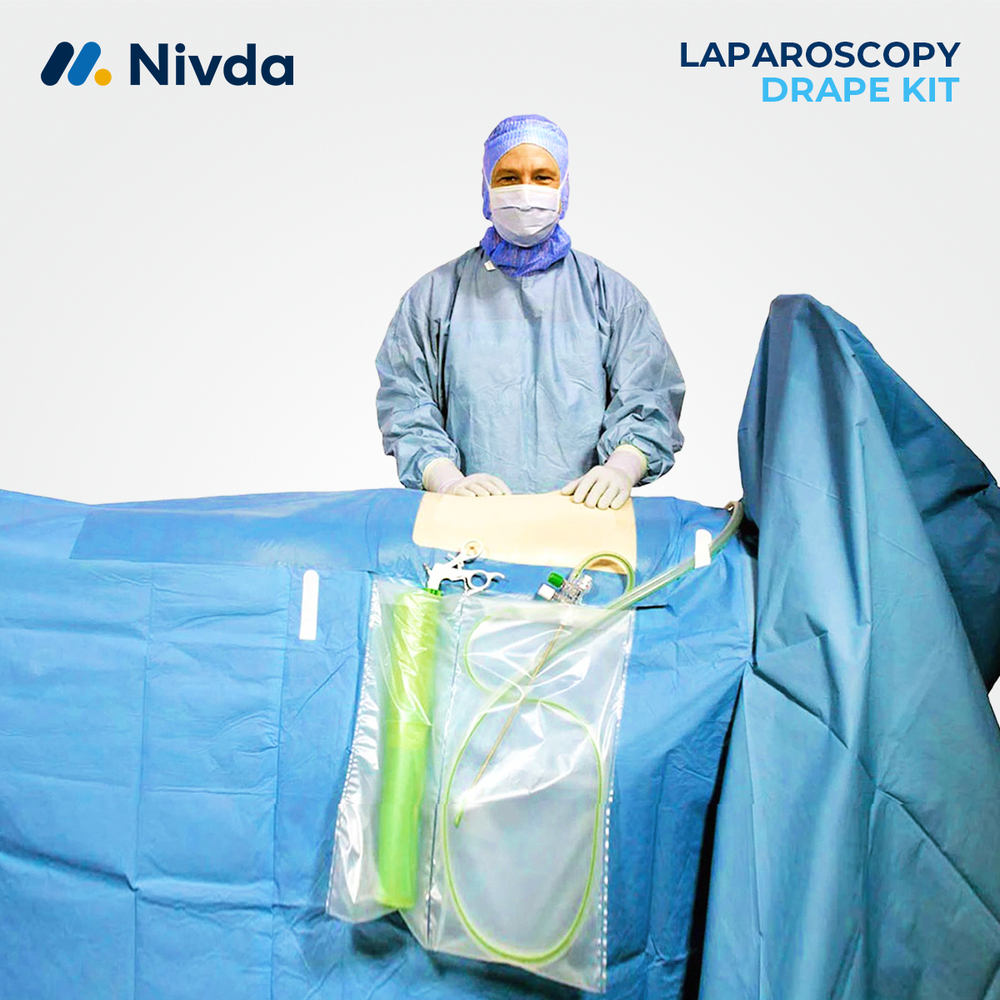
About Infection Prevention in Surgery
Nivda Drapes is globally leading manufacturer and supplier of Infection Prevention in Surgery. Infection Prevention in Surgery is a critical aspect of modern surgical practice, helping healthcare facilities maintain high standards of patient safety. Surgical drapes and disposable protective materials are used to isolate the operative site and limit the spread of microorganisms during procedures. Nivda Drapes provides reliable sterile draping solutions that support infection control protocols in hospitals and surgical centers. These products help maintain a protected surgical field, improve operating room efficiency, and contribute to safer surgical outcomes.Advanced Features:
Sterile disposable drapes designed for surgical infection control
Helps maintain a clean and protected surgical field
Fluid-resistant material to support barrier protection
Easy placement around the surgical site
Single-use design to support hygiene and safety
Suitable for hospitals, clinics, and surgical centers
الوقاية من العدوى ÙÙŠ الجراØØ©ØŒ مكاÙØØ© العدوى الجراØية، الأغطية والأردية المعقمة، وسائل الØماية الجراØية ذات الاستخدام الواØد، الوقاية من العدوى ÙÙŠ غر٠العمليات، منتجات الØواجز الطبية، المستلزمات الجراØية غير المنسوجة
Профилактика инфекций в хирургии, контроль хирургичеÑких инфекций, Ñтерильные проÑтыни и халаты, Ð¾Ð´Ð½Ð¾Ñ€Ð°Ð·Ð¾Ð²Ð°Ñ Ñ…Ð¸Ñ€ÑƒÑ€Ð³Ð¸Ñ‡ÐµÑÐºÐ°Ñ Ð·Ð°Ñ‰Ð¸Ñ‚Ð°, профилактика инфекций в операционной, медицинÑкие барьерные изделиÑ, нетканые хирургичеÑкие принадлежноÑти
Prevención de infecciones en cirugÃa, control de infecciones quirúrgicas, campos y batas estériles, protección quirúrgica desechable, prevención de infecciones en quirófano, productos de barrera médica, suministros quirúrgicos no tejidos
Prévention des infections en chirurgie, contrôle des infections chirurgicales, champs et blouses stériles, protections chirurgicales à usage unique, prévention des infections au bloc opératoire, produits de barrière médicale, fournitures chirurgicales non tissées
Comprehensive Infection Control
Focusing on every aspect of surgical site protection, this kit brings together essential barrier components. Items are engineered to reduce cross-contamination during surgery, adhering strictly to international sterilization and packaging standards. Surgeons and staff can trust this single-use kit for safe sterile field establishment.
Certified Safety and Quality
All components meet CE/ISO 13485 certification, confirming strict compliance with global medical device directives. The use of ethylene oxide sterilization and tamper-proof packaging further underscores the integrity and efficacy of this infection prevention kit in critical settings.
Convenient, Ready-to-Use Design
Thoughtfully assembled for immediate deployment, the kit is easily portable and designed for single use. Standard sizing (35 cm x 22 cm x 8 cm) and a weight of approximately 1.2 kg ensure straightforward transport and handling, making it ideal for swift preparation in surgical suites.
FAQ's of Infection Prevention in Surgery:
Q: How does ethylene oxide sterilization enhance infection prevention in this surgical kit?
A: Ethylene oxide sterilization ensures that all medical-grade components-such as drapes, gloves, and masks-are thoroughly free of microorganisms by penetrating and neutralizing pathogens, even in hard-to-reach areas. This ensures every item remains sterile until use, significantly reducing infection risks in the surgical environment.Q: What is included in the Infection Prevention in Surgery Kit, and how should it be used?
A: The kit includes sterile drapes, gloves, masks, and disinfectants, all made from latex-free, medical-grade materials. Intended for single-use only, the kit supports the preparation of a sterile field before and during surgery. Simply open the tamper-proof packaging just prior to the procedure, set up the items as per your operating room protocol, and dispose of everything after a single use.Q: When should this kit be used in the surgical process?
A: This kit is specifically designed for both preoperative and intraoperative phases. It should be unwrapped and utilized immediately before the surgical procedure to establish and maintain a sterile field throughout the surgery, helping to minimize contamination risks at every critical stage.Q: Where is this kit most suitable for application?
A: The Infection Prevention in Surgery Kit is intended for use in hospital and clinic surgery rooms. Its compact and portable design also enables easy handling within the operating suite, ensuring immediate access for all surgical team members.Q: What are the benefits of using a CE/ISO 13485 certified, single-use surgical barrier kit?
A: This certification ensures the kit complies with stringent international quality and safety standards, reducing the risk of healthcare-associated infections. Single-use, sterile, and latex-free components further minimize cross-contamination and allergen exposure, promoting patient and staff safety during surgical procedures.Q: How should the kit be stored, and what is its shelf life?
A: To maintain sterility, the kit should be stored in a cool, dry place away from direct sunlight. Each kit offers a shelf life of 2 years, provided it remains sealed and properly stored according to manufacturer instructions.






Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Disposable Drapes Category
Surgical Laparoscopy Pack
Price 7.23 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Operating Type : Other, Manual Use
Weight : Lightweight
Features : Waterproof, Tear Resistance, High Absorbency, Antistatic, Bacterial Barrier
Technology : Disposable Sterile Manufacturing
OR Perineal Sheet 80 x 100 cm
Price 0.50 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Operating Type : Other, Singleuse, Disposable
Weight : Lightweight
Features : Waterproof, Tearresistant, Lintfree, High Absorbency, Breathable
Technology : SMS (SpunbondMeltblownSpunbond)
Femoral Angiography Drape
Price 4.63 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Operating Type : Other, Manual Application
Weight : Lightweight, approximately 80120g per drape
Features : Fluid repellent, high absorbency, tear resistant, sterile barrier, fenestration for catheter insertion
Technology : Sterile, Disposable
Reinforced Surgical Gowns
Price 3.59 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Operating Type : Other, Disposable, Manual Donning
Weight : Lightweight (approx. 100150g per gown)
Features : Fluid Resistance, Tear Resistant, Breathable Back Panel, Reinforced Critical Zones, Sterile
Technology : Ultrasonic Sealed Seams

 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry



 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese