Pacemaker Drape
Price 5.21 USD ($)/ Piece
Pacemaker Drape Specification
- Storage Instructions
- Store in a dry, cool place away from direct sunlight
- Function
- Creates sterile environment; covers patient except for operating site
- Usage Type
- Medical/Hospital
- Instruments Type
- Surgical Drape
- Features
- Impervious, Tear-resistant, Fluid Control, Adhesive Area for Secure Placement
- Shelf Life
- Up to 5 Years (Unopened)
- Equipment Type
- Pacemaker Drape
- Material
- Sterile Medical Grade SMS Fabric
- Condition
- New, Sterile
- Technology
- Nonwoven Fabric Technology
- Portable
- Yes
- Wall Mounted
- No
- Noise Level
- Silent
- Operating Type
- Disposable, Single-Use
- Use
- Used to create a sterile barrier during pacemaker or device implant procedures
- Dimension (L*W*H)
- 180 cm x 150 cm (Standard)
- Weight
- Lightweight
- Color
- Blue
- Product Code
- PMD-180150
- Compatibility
- Suitable for All Pacemaker Insertion Procedures
- Fluid Collection Pouch
- Integrated
- Adhesive Area Size
- 8 x 12 inches (for instrument site fixation)
- Packaging Type
- Individual Peel Pouch
- Latex Content
- Latex Free
- Sterility Assurance Level
- 10
- Edge Type
- Ultrasonically Sealed Edges
- Transparency
- Opaque
- Compliance
- CE and ISO 13485 Certified
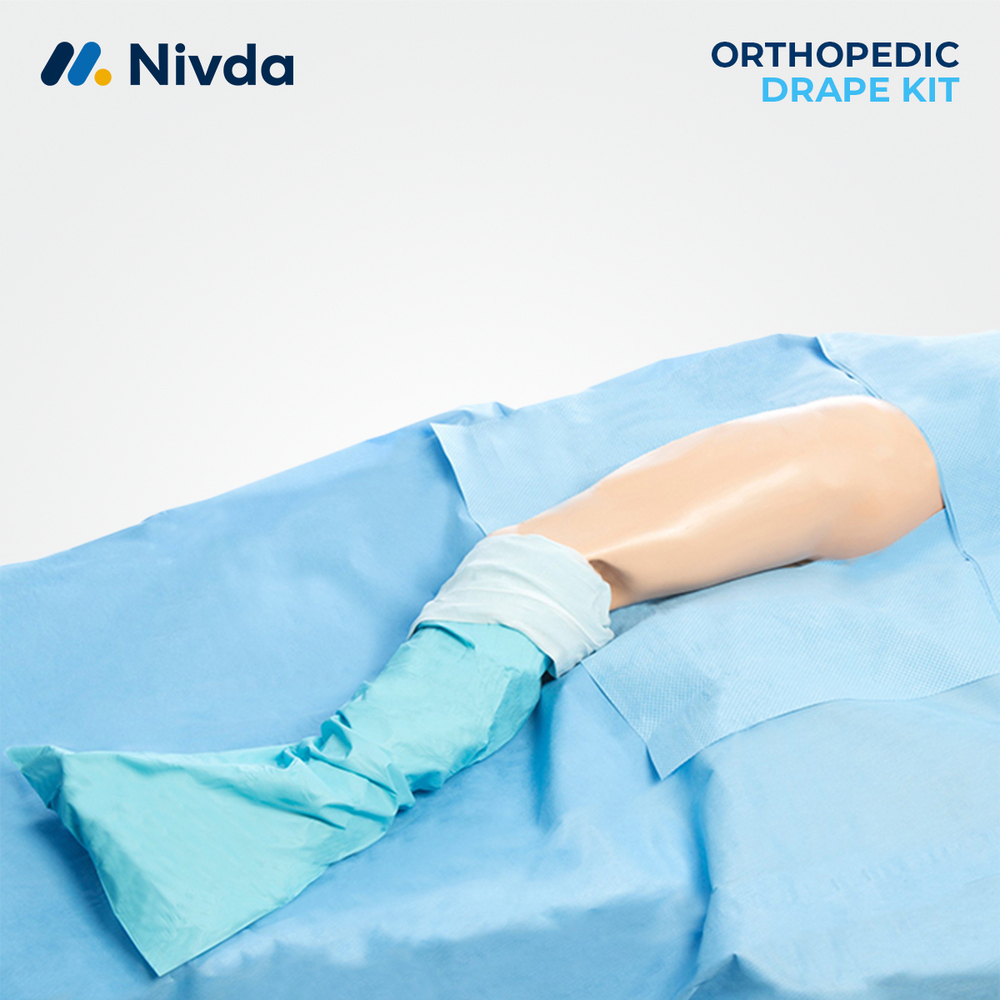
About Pacemaker Drape
The Pacemaker Drape plays an important role in infection control during cardiac device implantation procedures. Its design allows precise exposure of the implantation site while supporting organized draping and fluid control. The drape helps medical teams maintain aseptic conditions and smooth workflow throughout the procedure. Nivda Drapes offers this disposable drape as a reliable solution that meets hospital standards for safety, hygiene, and consistency in cardiac care environments.
Advanced Features
Designed specifically for pacemaker implantation proceduresHelps maintain a sterile and organized operative field
Supports fluid control during cardiac interventions
Suitable for hospitals and cardiac procedure rooms
Pacemaker Drape, Cardiac Surgical Drape, Pacemaker Implant Drape, Sterile Cardiac Drape, Disposable Surgical Drape, OT Cardiac Drape, Non Woven Surgical Drape
غطاء جهاز تنظيم ضربات القلب، غطاء جراØØ© القلب، غطاء زرع جهاز تنظيم ضربات القلب، غطاء معقم للقلب، غطاء جراØÙŠ للاستخدام مرة واØدة، غطاء غرÙØ© عمليات القلب، غطاء جراØÙŠ غير منسوج
ХирургичеÑÐºÐ°Ñ Ð¿Ñ€Ð¾ÑÑ‚Ñ‹Ð½Ñ Ð´Ð»Ñ ÐºÐ°Ñ€Ð´Ð¸Ð¾ÑтимулÑтора, проÑÑ‚Ñ‹Ð½Ñ Ð´Ð»Ñ Ð¸Ð¼Ð¿Ð»Ð°Ð½Ñ‚Ð°Ñ†Ð¸Ð¸ кардиоÑтимулÑтора, ÑÑ‚ÐµÑ€Ð¸Ð»ÑŒÐ½Ð°Ñ Ñ…Ð¸Ñ€ÑƒÑ€Ð³Ð¸Ñ‡ÐµÑÐºÐ°Ñ Ð¿Ñ€Ð¾ÑÑ‚Ñ‹Ð½Ñ Ð´Ð»Ñ Ñердца, Ð¾Ð´Ð½Ð¾Ñ€Ð°Ð·Ð¾Ð²Ð°Ñ Ñ…Ð¸Ñ€ÑƒÑ€Ð³Ð¸Ñ‡ÐµÑÐºÐ°Ñ Ð¿Ñ€Ð¾ÑтынÑ, хирургичеÑÐºÐ°Ñ Ð¿Ñ€Ð¾ÑÑ‚Ñ‹Ð½Ñ Ð´Ð»Ñ Ð¾Ð¿ÐµÑ€Ð°Ñ†Ð¸Ð¾Ð½Ð½Ð¾Ð¹, Ð½ÐµÑ‚ÐºÐ°Ð½Ð°Ñ Ñ…Ð¸Ñ€ÑƒÑ€Ð³Ð¸Ñ‡ÐµÑÐºÐ°Ñ Ð¿Ñ€Ð¾ÑтынÑ
Campo quirúrgico para marcapasos, campo quirúrgico cardÃaco, campo quirúrgico para implante de marcapasos, campo quirúrgico cardÃaco estéril, campo quirúrgico desechable, campo quirúrgico cardÃaco para quirófano, campo quirúrgico no tejido
Champ opératoire pour stimulateur cardiaque, champ opératoire pour chirurgie cardiaque, champ opératoire pour implantation de stimulateur cardiaque, champ stérile pour chirurgie cardiaque, champ opératoire jetable, champ opératoire pour chirurgie cardiaque, champ opératoire non tissé
Superior Barrier for Sterile Procedures
Engineered with sterile, medical-grade SMS fabric and nonwoven technology, this pacemaker drape guarantees a sterile field and is designed for all pacemaker insertion procedures. Its impervious, tear-resistant material and ultrasonically sealed edges enhance patient safety by effectively preventing microorganism transfer and fluid seepage.
Integrated Features for Enhanced Performance
The drape includes an integrated fluid collection pouch for efficient fluid control, a strategically sized adhesive area (8 x 12 inches) for secure placement, and is entirely latex-free to minimize allergic reactions. Its lightweight, portable design ensures easy setup and removal, supporting optimal workflow during device implant surgeries.
Compliance and Reliable Sterility
Fully compliant with CE and ISO 13485 certifications, this single-use, disposable drape is individually peel-packed for assured sterility (SAL 106). With a shelf life of up to five years, it provides long-term reliability and performance in both medical and hospital settings.
FAQ's of Pacemaker Drape:
Q: How does the pacemaker drape maintain a sterile environment during procedures?
A: The pacemaker drape creates a sterile barrier using medical-grade SMS fabric and ultrasonically sealed edges. It is designed for single use and individually peel-packed to ensure sterility with a Sterility Assurance Level of 106, effectively preventing contamination during pacemaker or device implant procedures.Q: What are the benefits of the integrated fluid collection pouch and adhesive area?
A: The integrated fluid collection pouch manages surgical fluids efficiently, keeping the surgical field dry, while the 8 x 12-inch adhesive area secures the drape in place around the instrument site. These features improve visibility, safety, and overall workflow during pacemaker insertion procedures.Q: When should the pacemaker drape be used and in what settings?
A: This drape is specifically intended for use during pacemaker insertion and related device implantation surgeries. It is suitable for medical and hospital environments where a sterile barrier and effective fluid management are crucial.Q: Where should the pacemaker drape be stored to maintain sterility and shelf life?
A: Store the drape in a dry, cool place away from direct sunlight. When kept unopened and in appropriate conditions, it retains its sterility and performance for up to five years from the date of manufacture.Q: What is the process for using the pacemaker drape during a procedure?
A: To use, remove the drape from its sterile individual peel pouch, position it to cover the patient while exposing the operative site, secure the adhesive area around the instrument site, and ensure the fluid collection pouch is correctly placed. Dispose of the drape after single use to maintain infection control.Q: Is the drape suitable for patients or staff sensitive to latex?
A: Yes, the pacemaker drape is entirely latex-free, making it safe for use with individuals who have latex sensitivities or allergies.











Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Surgical Drapes Category
Advin Orthopaedic Drapes
Price 4 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Instruments Type : Drape/consumable
Features : Fluid repellent, lint free, tear resistant, good drapability
Technology : Ultrasonically sealed edges
Shelf Life : 5 years from date of manufacture
Joint Replacement Drape Kit
Price 7.48 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Instruments Type : Surgical Drape Kit
Features : Fluid Absorbent, Lint Free, Tear Resistant, Impervious to Fluids, Infection Barrier, EO Sterilized
Technology : SMS/SSMMS Technology
Shelf Life : Up to 3 Years
Disposable Pcnl Drapes
Price 3.01 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Instruments Type : Surgical Drapes
Features : Adhesive Incise Area, Fluid Collecting Pouch, Soft, Lint Free, SplashProof, Hydrophobic, Tear Resistant, FiberFree Filtration
Technology : Ultrasonic Sealed & Reinforced
Shelf Life : 5 Years from date of manufacture

 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry



 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese