Peripheral Embolectomy Catheter
Price 30 USD ($)/ Piece
Peripheral Embolectomy Catheter Specification
- Storage Instructions
- Store in a cool, dry place away from direct sunlight
- Measurement Range
- Balloon size: 2F to 8F
- Features
- Soft, atraumatic balloon tip; radiopaque marker; multiple sizes available; smooth shaft
- Accuracy
- Balloon inflation, accurate to catheter lumen
- Usage Type
- Single use only
- Shelf Life
- 3-5 years
- Instruments Type
- Catheter
- Function
- Embolus and thrombus extraction
- Equipment Type
- Peripheral Embolectomy Catheter
- Material
- Medical grade PVC / Polyurethane
- Condition
- Sterile
- Technology
- Balloon embolectomy
- Portable
- Yes
- Wall Mounted
- No
- Real-Time Operation
- Yes
- Noise Level
- Silent
- Operating Type
- Manual
- Use
- Removal of emboli and thrombi from peripheral arteries and veins
- Dimension (L*W*H)
- Varies by size (commonly 40-80 cm length)
- Weight
- Lightweight, < 50g
- Color
- Transparent/Clear with blue tip
- Intended Use
- Peripheral vascular intervention
- Packaging
- Individually sterile packed
- Sterilization Method
- Ethylene oxide (ETO)
- Tip Type
- Soft rounded/atraumatic tip
- Size Available
- 2F, 3F, 4F, 5F, 6F, 7F, 8F
- Balloon Material
- Latex or silicone rubber
- Connector Type
- Luer lock connector for syringe attachment
- Radiopacity
- Radiopaque marker at distal tip for fluoroscopic visualization
- Balloon Length
- Varies (typically 10-15 mm)
- Compliance Standard
- Meets ISO and CE standards for medical devices
- Latex Content
- Latex or latex-free variants available
About Peripheral Embolectomy Catheter
The peripheral embolectomy catheter is designed to support effective clot removal while preserving vessel integrity during peripheral vascular procedures. Its balloon-assisted mechanism allows controlled engagement and retrieval of emboli to help restore normal blood flow. Advin Health Care manufactures this catheter with a strong emphasis on quality, safety, and consistent clinical performance. The catheter is suitable for use in a wide range of peripheral arterial applications, supporting efficient workflow and reliable clinical outcomes.
Advanced Features-
Inflatable balloon for controlled embolus removal
-
Smooth catheter shaft for easy navigation in peripheral vessels
-
Designed to support restoration of blood flow
-
Intended for single-use clinical applications
Product Keywords
Peripheral Embolectomy Catheter, Embolectomy Balloon Catheter, Peripheral Thrombectomy Catheter, Clot Removal Catheter, Vascular Embolectomy Device, Balloon Catheter for Embolus Removal
U3 3aU U UUUU U3 UUU 3aU U U3 3aU U UUUU U3 2U UU U2 3aU U UU1U U3 UUU U2U U
NNN N N NN NNo341 N1434NoN3414 , 341212N1 oNNN N N1434NoN3414 , oNNN N N NN NNo341 NN3414NoN3414 , oNNN N N 12 N NN3414342, NNNN341NN234 N N34NN NN341 N1434NoN3414 , 341212N1 oNNN N N 12 N N1434342.
CatAter de embolectomAa perifArica, catAter de balA3n para embolectomAa, catAter de trombectomAa perifArica, catAter para extracciA3n de coAgulos, dispositivo de embolectomAa vascular, catAter de balA3n para extracciA3n de Ambolos
CathAter d'embolectomie pAriphArique, cathAter A ballonnet d'embolectomie, cathAter de thrombectomie pAriphArique, cathAter d'extraction de caillot, dispositif d'embolectomie vasculaire, cathAter A ballonnet pour l'extraction d'emboles
Precision-Controlled Vascular Intervention
Engineered for real-time, manual operation, the Peripheral Embolectomy Catheter ensures accurate balloon inflation and extraction of emboli or thrombi. The soft rounded tip provides an atraumatic approach, while its radiopaque marker offers clear visualization under fluoroscopy for heightened procedural control. With smooth shaft construction and various size options, clinicians can select the best fit for individual patient needs.
Tailored to Clinical Demands
Available in multiple sizes (2F-8F) and balloon lengths, this catheter supports a wide range of peripheral vascular interventions. Medical-grade PVC or polyurethane offers a balance of flexibility and strength, while latex or latex-free balloon options cater to diverse patient sensitivities. The lightweight design simplifies handling and optimizes patient safety during removal procedures.
Enhanced Sterility and Compliance
Each catheter is sterilized using ethylene oxide (ETO) and packaged individually, promoting infection control and procedural efficiency. The product meets stringent ISO and CE standards, verifying its adherence to global safety and quality guidelines. Storage is straightforward-keep in a cool, dry environment away from direct sunlight for up to 3-5 years.
FAQ's of Peripheral Embolectomy Catheter:
Q: How is the Peripheral Embolectomy Catheter used during procedures?
A: The catheter is manually operated by inserting the balloon-tipped device into peripheral arteries or veins, inflating the balloon to capture emboli or thrombi, and then withdrawing the catheter to remove obstructions safely. The radiopaque marker assists clinicians in tracking catheter position through fluoroscopy.Q: What sizes and variant options does the catheter offer?
A: The device is available in sizes ranging from 2F to 8F with balloon lengths typically between 10-15 mm. Balloon materials include latex and silicone rubber, with both latex and latex-free versions provided to address patient allergies.Q: When should this catheter be selected for vascular intervention?
A: It is appropriate for patients requiring peripheral vascular intervention, specifically for extracting emboli or thrombi from peripheral arteries or veins. Its soft, atraumatic tip and precise visualization capabilities make it suitable for delicate vessel navigation.Q: Where do the safety and compliance standards come into play?
A: Manufactured by certified suppliers in India, the catheter adheres to ISO and CE standards, ensuring it meets stringent global requirements for safety and quality in clinical use.Q: What is the process for ensuring the catheter's sterility and proper storage?
A: Each unit is sterilized using ethylene oxide (ETO) and individually packed to maintain sterility. It should be stored in a cool, dry place away from direct sunlight, ensuring a shelf life of up to 3-5 years.Q: What benefits does this catheter provide in clinical practice?
A: The device's soft, atraumatic tip minimizes vessel trauma, while its wide range of sizes and material options allow tailored patient care. The luer lock connector and radiopaque marker further enhance procedural safety and efficiency.Q: How precise is the balloon inflation system on the catheter?
A: Balloon inflation is engineered for accuracy relative to the catheter lumen, aiding in effective embolus or thrombus extraction while maintaining safety throughout the procedure.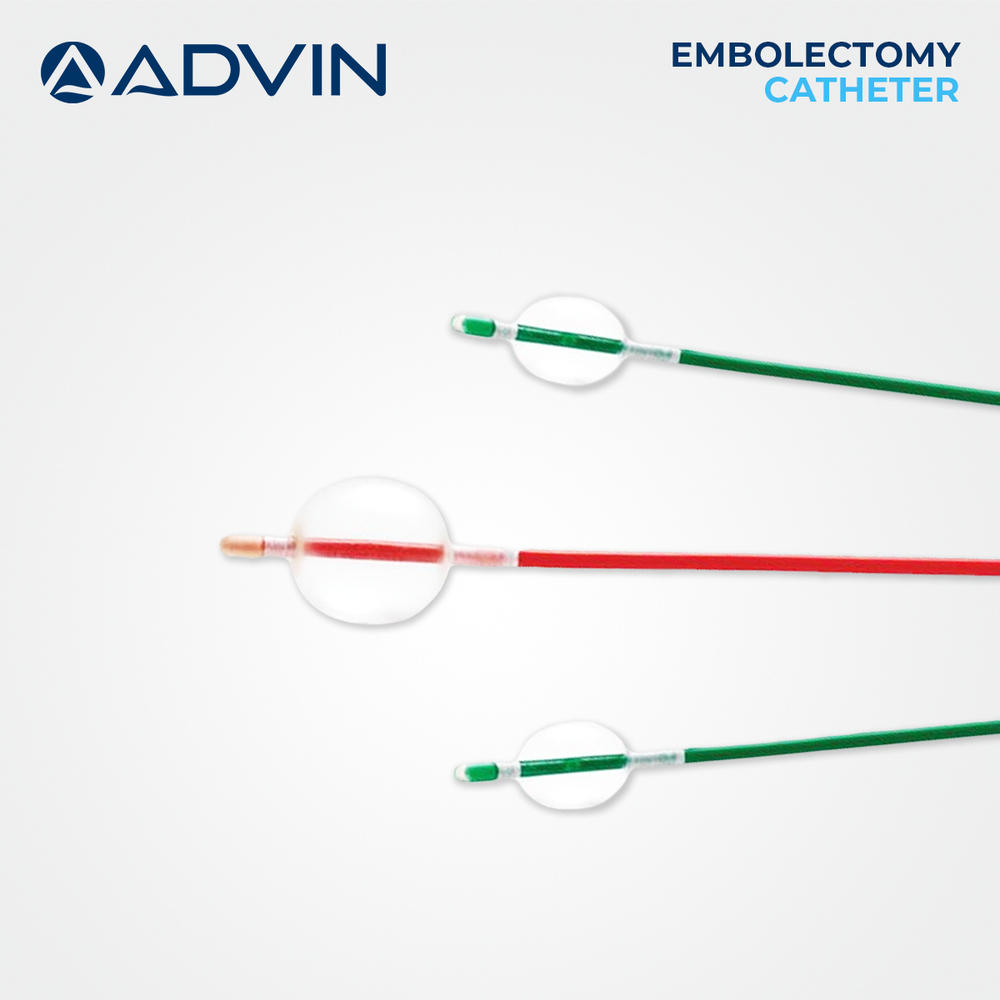


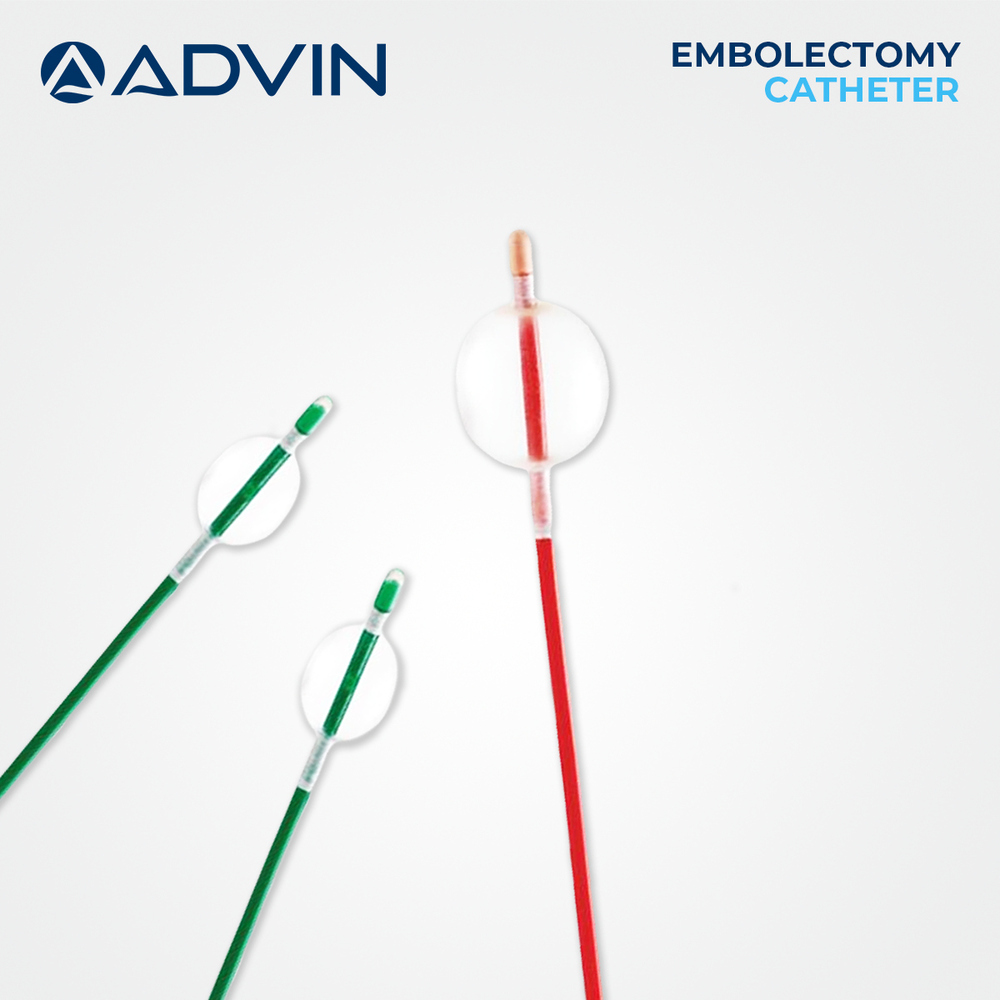
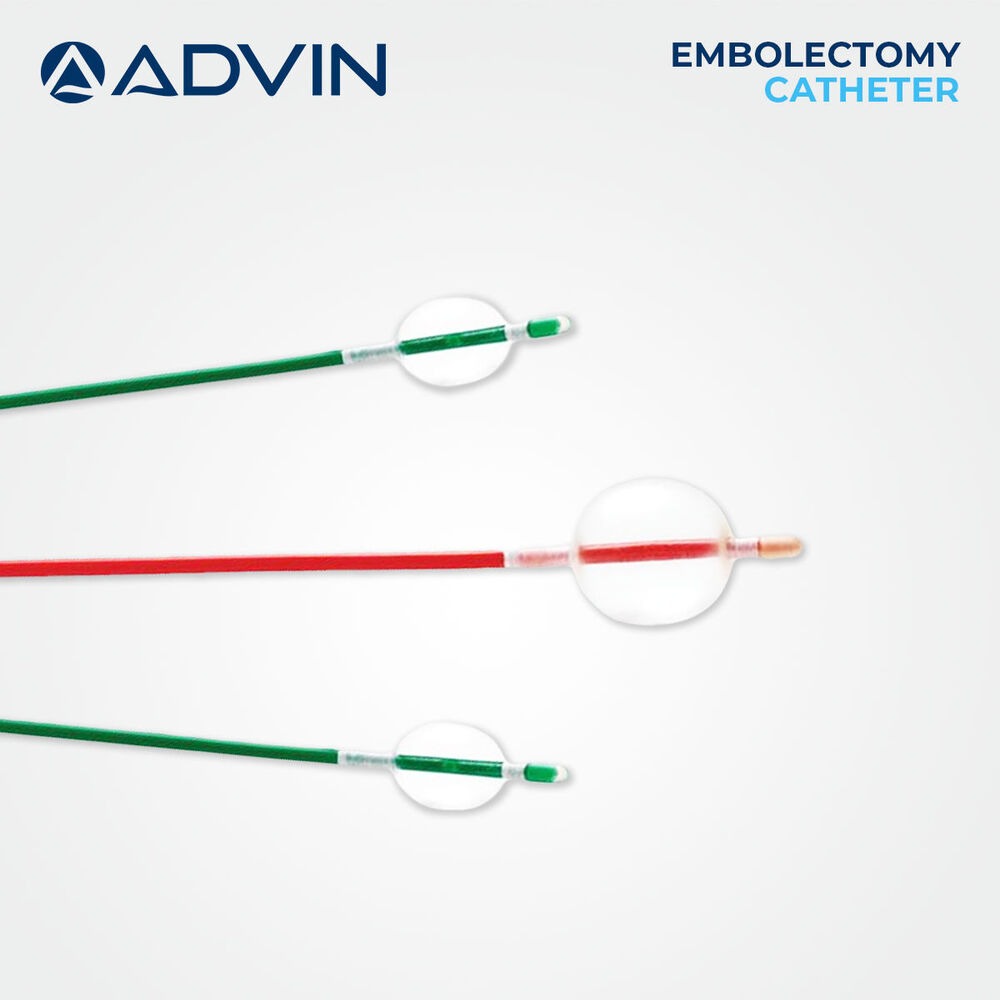
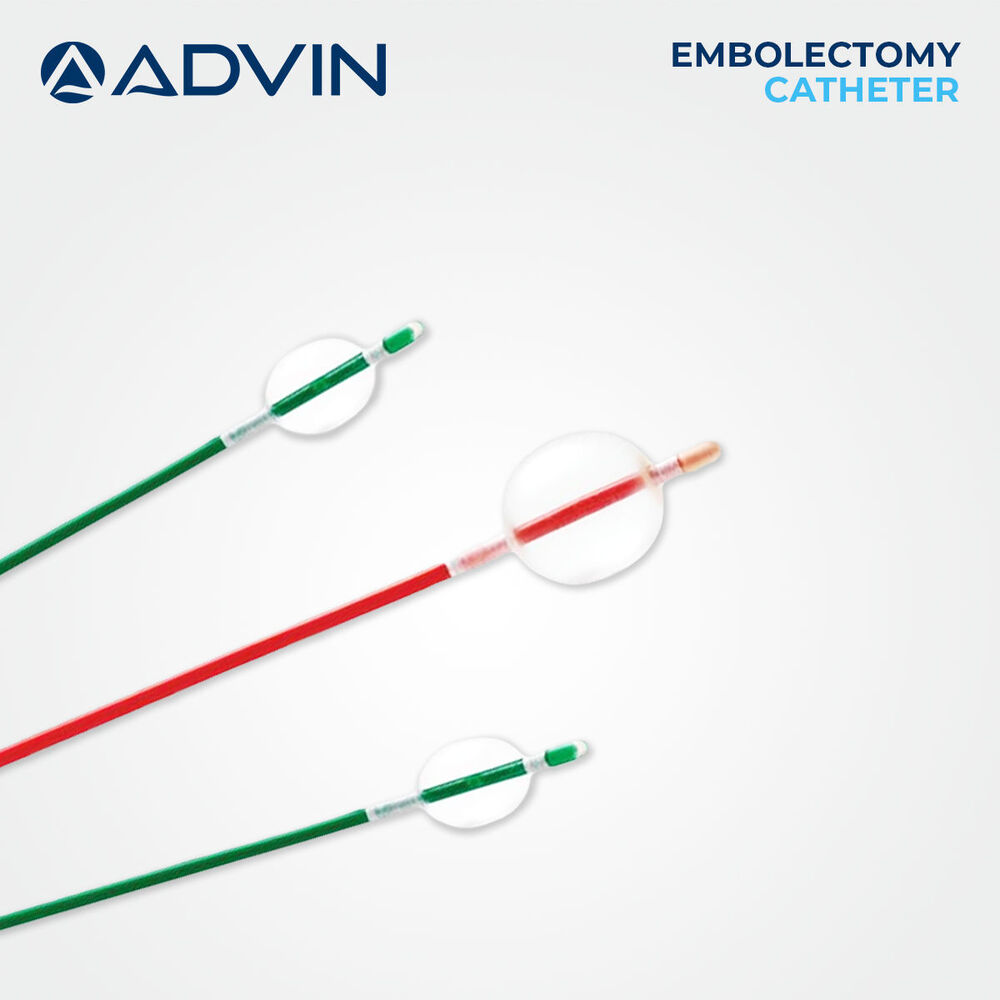

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+

 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry


 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese