Spinal Fixation System
Price 1 USD ($)/ Piece
Spinal Fixation System Specification
- Function
- Stabilization of the spine post-surgery
- Shelf Life
- 5 Years (Unopened in sterile packaging)
- Usage Type
- Hospital and Surgical Center Use
- Storage Instructions
- Store in cool, dry place away from direct sunlight
- Measurement Range
- Available in various lengths and diameters
- Accuracy
- High precision fit
- Instruments Type
- Orthopedic Surgical Implant
- Features
- Biocompatible, Corrosion Resistant, Radiolucent Markers, Versatile Fixation Options
- Equipment Type
- Spinal Fixation System
- Material
- Titanium Alloy
- Condition
- New
- Technology
- Orthopedic Implant Technology
- Portable
- Yes
- Wall Mounted
- No
- Real-Time Operation
- Yes
- Noise Level
- Silent Operation
- Operating Type
- Manual
- Use
- Spinal Fusion and Stabilization Surgery
- Dimension (L*W*H)
- Custom sizes available (varies with patient requirements)
- Weight
- Lightweight (Implant Grade)
- Color
- Metallic / Silver
- Radiology Compatibility
- MRI and CT Compatible
- Fixation Levels
- Multi-level fixation possible
- Regulatory Compliance
- ISO 13485, CE Marked
- Screw Type Compatibility
- Polyaxial and Monoaxial Pedicle Screws
- Rod Diameter Options
- Typically 5.5mm and 6.0mm
- Application
- Thoracic, Lumbar, Cervical Spine
- Packaging Type
- Sterile Double Blister Pack
- Sterilization Method
- Gamma Radiation/EO Sterilized
- System Components
- Pedicle Screws, Rods, Connectors, Set Screws
About Spinal Fixation System
Spinal Fixation System is widely used in trauma orthopedic surgeries for spinal fractures, deformities, and degenerative conditions. The system provides stable support to vertebrae, enabling precise alignment and promoting spinal fusion. Advin Health Care manufactures Spinal Fixation Systems with premium medical-grade materials, ensuring biocompatibility, long-term durability, and consistent performance. Available in various configurations, including rods, screws, and plates, it accommodates different patient anatomies and surgical techniques, offering optimal clinical outcomes and improved spinal stability.
Advanced Features:
-
Made from high-quality medical-grade materials for strength, durability, and biocompatibility
-
Provides stable spinal support and facilitates vertebral alignment
-
Modular system with rods, screws, and plates for versatile surgical applications
-
Helps reduce pain and restore spinal function in trauma and degenerative conditions
-
Suitable for spinal fracture stabilization, deformity correction, and reconstructive surgeries
Versatile Fixation for Spinal Stability
The Spinal Fixation System supports flexible application in thoracic, lumbar, and cervical surgeries, enabling multi-level fixation as required by patient-specific needs. Its compatibility with both polyaxial and monoaxial pedicle screws and availability of different rod diameters offer the surgeon adaptability during procedures. Advanced biocompatible materials ensure safety and long-term performance.
Advanced Sterile Packaging and Compliance
Each system is sterilized using either gamma radiation or EO and presented in a double blister pack, ensuring optimal sterility up to five years if unopened. The implants are manufactured under ISO 13485 standards and hold CE marking, confirming their quality, safety, and adherence to global regulatory benchmarks.
Superior Imaging Compatibility
Radiolucent markers and titanium alloy construction make the system fully compatible with MRI and CT scans, providing clear postoperative imaging for assessment and follow-up. The design is intended for seamless integration into modern surgical practice while maintaining patient safety and comfort.
FAQ's of Spinal Fixation System:
Q: How is the Spinal Fixation System sterilized before use?
A: The system undergoes sterilization using either gamma radiation or ethylene oxide (EO) processes. Each component is then packed in a sterile double blister pack for assured protection and maintained sterility for up to five years when unopened.Q: What types of screws and rod diameters does the system support?
A: This system is compatible with both polyaxial and monoaxial pedicle screws, offering flexibility during complex surgeries. Rod diameter options typically include 5.5mm and 6.0mm, accommodating a range of patient anatomies and fixation requirements.Q: When can the Spinal Fixation System be used in surgical procedures?
A: It is indicated for use during spinal fusion and stabilization surgeries involving the thoracic, lumbar, or cervical spine, particularly where multi-level fixation is necessary for optimal spinal alignment and healing support.Q: Where should the Spinal Fixation System be stored prior to use?
A: The product must be stored in a cool, dry location away from direct sunlight to preserve sterile integrity and maximize shelf life. Avoiding temperature extremes ensures component longevity and safety.Q: What is the process for selecting the correct size for implantation?
A: The system offers a variety of lengths and diameters for rods and screws, allowing the surgeon to select the optimal size tailored to each patient's anatomical and surgical needs. Custom dimensions can also be supplied when required for unique cases.Q: How does the system benefit postoperative imaging and assessment?
A: Constructed from titanium alloy with radiolucent markers, the system offers excellent MRI and CT compatibility. This facilitates clear, unobstructed imaging for accurate postoperative assessment without interference from metallic artifacts.Q: What are the main benefits of using this spinal fixation system?
A: Key advantages include high biocompatibility, precise fit, lightweight design, versatile fixation options, corrosion resistance, and silent, manual operation. Its well-validated performance helps ensure safe and effective spinal stabilization in hospital and surgical center settings.






Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Orthopedic Products Category
All Suture Anchor with 2 pc Fiber
Price 26 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Usage Type : Surgical/medical
Storage Instructions : Store in a cool, dry place, away from direct sunlight
Function : Soft tissue anchoring, suture fixation to bone
Shelf Life : 35 years (unopened)
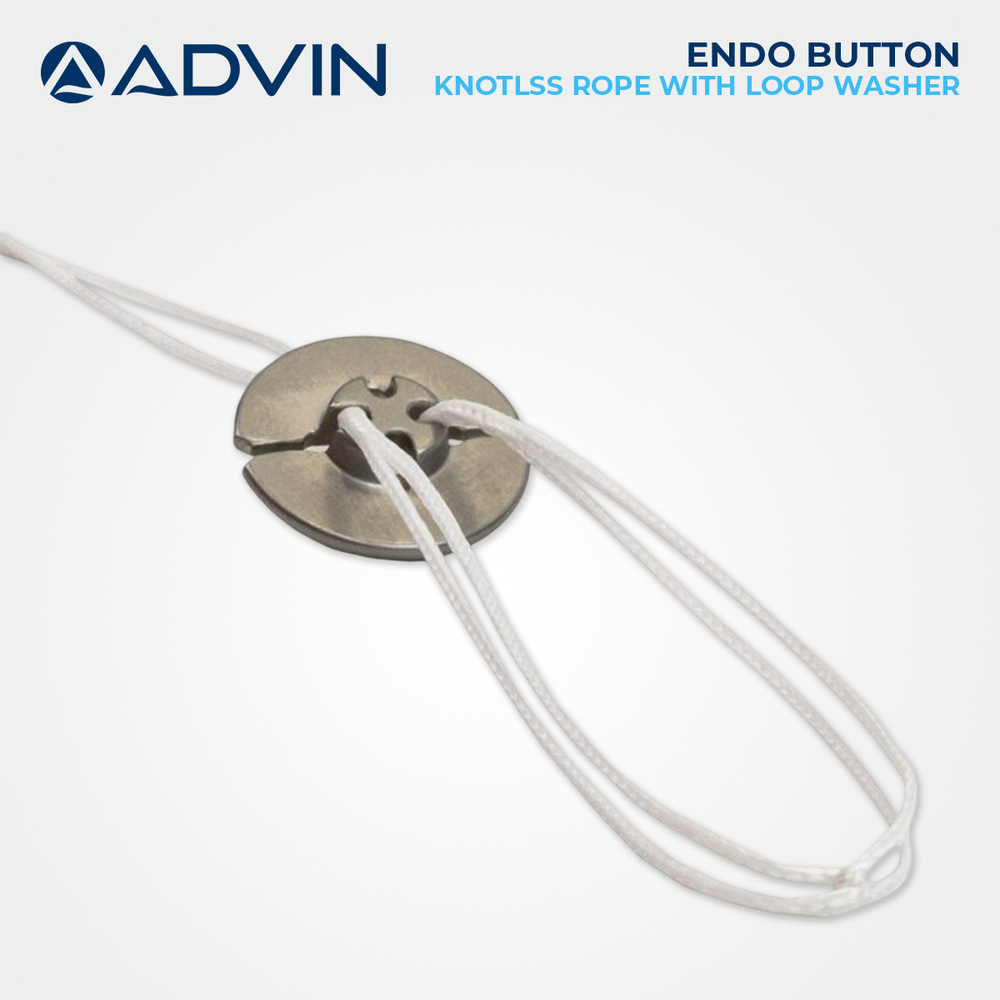
Endo Button knotlss Rope with Loop washer
Price 10 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Usage Type : Medical / Surgical
Storage Instructions : Store in Cool, Dry Place. Keep Sterile Until Use.
Function : Fixation and Stabilization of Soft Tissue to Bone
Shelf Life : 5 Years (Sterile Packaging)
All-Metal FiberFix Suture Anchor Single Fiber
Price 400 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Usage Type : Single Use
Storage Instructions : Store in a Cool, Dry Place in Original Packaging
Function : Provides secure soft tissue reattachment to bone
Shelf Life : 5 Years
T Buttress Plate
Price 5 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Usage Type : Single use
Storage Instructions : Store in dry, sterile conditions
Function : Bone stabilization and fixation during orthopedic surgery
Shelf Life : 5 Years

 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry



 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese