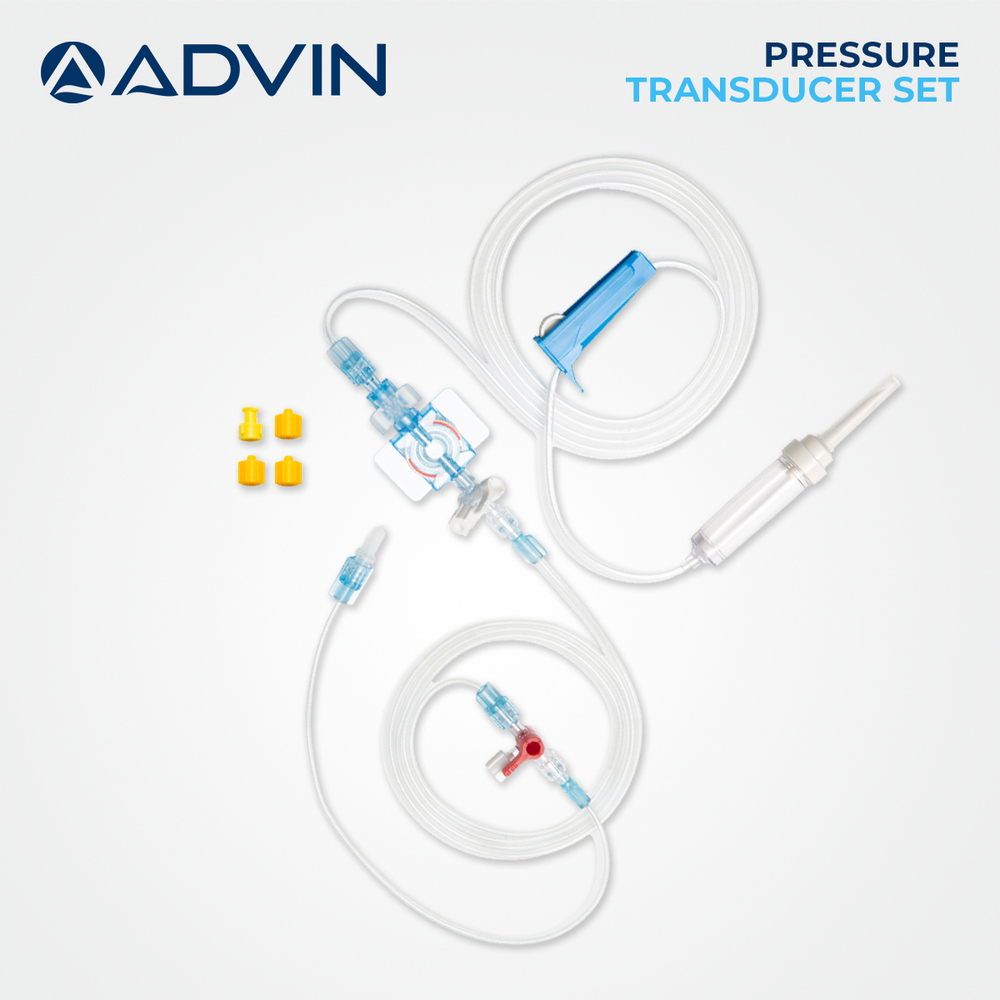
Sterile Pressure Transducer
Price 9.7 USD ($)/ Piece
Sterile Pressure Transducer Specification
- Instruments Type
- Medical Diagnostic Device
- Usage Type
- Single Use
- Accuracy
- 1 mmHg
- Frequency
- Monitor Dependent
- Shelf Life
- Up to 5 years (if unopened)
- Function
- Real-time invasive blood pressure measurement
- Display Type
- Monitor Dependent
- Measurement Range
- -50 to +300 mmHg
- Features
- Disposable, Sterile, Latex-free, Fluid path separation
- Storage Instructions
- Store in a cool, dry place. Avoid direct sunlight.
- Equipment Type
- Sterile Pressure Transducer
- Material
- Medical-grade Polycarbonate and Silicone
- Condition
- New, Sterile
- Technology
- Disposable MEMS Sensor
- Portable
- Yes
- Wall Mounted
- No
- Real-Time Operation
- Yes
- Noise Level
- Low
- Operating Type
- Manual and Compatible with Electronic Monitors
- Use
- Medical Pressure Monitoring
- Power Source
- External (Monitor Supplied)
- Power Consumption
- Negligible
- Dimension (L*W*H)
- Standard: 90 x 35 x 20 mm
- Weight
- Approx. 45 g
- Color
- Transparent with Blue Accents
- Fluid Path
- Non-DEHP, Non-Pyrogenic
- Mounting Options
- With Detachable Plate Adapter
- Disposability
- Single-use, Disposable
- Packaging
- Individually Blister Packed
- Application Areas
- ICU, CCU, Operation Theater, Cardiac Unit
- Tubing Length
- 100 cm (customizable)
- Compliance
- CE Marked, ISO 13485 Certified
- Operating Temperature
- 0C to 45C
- Sterilization Method
- Ethylene Oxide (EO) Sterilized
- Connector Type
- Standard Luer Lock
About Sterile Pressure Transducer
Advin Health Care is a trusted manufacturer of Sterile Pressure Transducers designed to meet international quality standards. These devices are engineered for accurate and reliable conversion of physiological pressure into electrical signals, ensuring precise monitoring in critical care, ICU, and catheterization lab environments with enhanced patient safety.
Key Features
- Pre-sterilized for immediate clinical use
- High-accuracy pressure measurement for reliable monitoring
- Stable signal output for consistent readings
- Fast response time for real-time pressure tracking
- Compatible with standard patient monitoring systems
- Single-use design to minimize infection risk
Specifications
- Type: Sterile Pressure Transducer
- Function: Pressure-to-electrical signal conversion
- Material: Medical-grade components
- Sterilization: EO Sterilized
- Usage: Single-use / Disposable
- Application: Invasive blood pressure (IBP), arterial, and venous pressure monitoring
Certifications
CE, ISO 13485, and ISO 9001 certified
Product Keywords
Sterile Pressure Transducer, IBP Transducer, Disposable Pressure Sensor, Blood Pressure Transducer, ICU Monitoring Device
, , , ,
Transductor de presin estril, transductor de PAI, sensor de presin desechable, transductor de presin arterial, dispositivo de monitorizacin de UCI
Transducteur de pression strile, transducteur de pression artrielle invasive, capteur de pression jetable, transducteur de pression artrielle, dispositif de surveillance en soins intensifs
Unmatched Sterility and Safety
Each Sterile Pressure Transducer undergoes EO sterilization and is packaged individually in blister packs to ensure absolute sterility and prevent cross-contamination. It is engineered from medical-grade polycarbonate and silicone, completely devoid of latex, DEHP, or pyrogens. Adherence to international quality standards, including CE Mark and ISO 13485 certification, guarantees both safety and reliability in every use.
Precise, Real-Time Blood Pressure Monitoring
Utilizing a disposable MEMS sensor, this device delivers highly accurate (1 mmHg) invasive blood pressure readings in real time. Its robust design supports both manual and electronic monitor compatibility, enabling seamless integration into ICU, CCU, operating rooms, and cardiac units. The standard Luer Lock connector and detachable plate adapter make setup straightforward and efficient.
Versatile Application and Customization
The Sterile Pressure Transducer's 100 cm tubing length, with customizable options, supports various procedural requirements. Its transparent construction with blue accents allows for easy fluid visualization, and the unit's lightweight, portable form ensures ease of movement. The device is intended for single-use, minimizing infection risk and promoting best practices across diverse care environments.
FAQ's of Sterile Pressure Transducer:
Q: How should the Sterile Pressure Transducer be stored before use?
A: Store the device in a cool, dry place, away from direct sunlight, to maintain its sterility and functionality until the expiration date. Keep the blister packaging unopened until ready for use.Q: What sterilization method is used for this transducer and why?
A: Ethylene Oxide (EO) sterilization is used for the pressure transducer, ensuring high-level sterilization without degrading medical-grade polycarbonate and silicone materials, making it safe and effective for sensitive patient applications.Q: When and where is this device typically used?
A: The Sterile Pressure Transducer is primarily used for real-time invasive blood pressure monitoring in intensive care units (ICU), cardiac care units (CCU), operation theaters, and cardiac units, especially where continuous, accurate monitoring is crucial.Q: What are the benefits of the disposable MEMS sensor technology in this transducer?
A: Disposable MEMS sensor technology provides accurate measurements (1 mmHg), reduces the risk of cross-infection, and ensures consistency for every new procedure due to its single-use, sterile configuration.Q: How is the fluid path designed to ensure patient safety?
A: The fluid path is completely non-DEHP and non-pyrogenic, minimizing the risk of adverse reactions. Its latex-free design further ensures compatibility with sensitive patients, and the sterile, sealed construction maintains a contamination-free interface.Q: Can the tubing length or connectors be customized?
A: Yes, while the standard tubing length is 100 cm, customization options are available on request. The device is equipped with a standard Luer Lock connector, ensuring compatibility with widely used monitoring systems.Q: What is the shelf life of the device, and can it be reused?
A: The device has a shelf life of up to 5 years if unopened and stored correctly. It is strictly intended for single-use only and should never be reused, as this can compromise sterility and patient safety.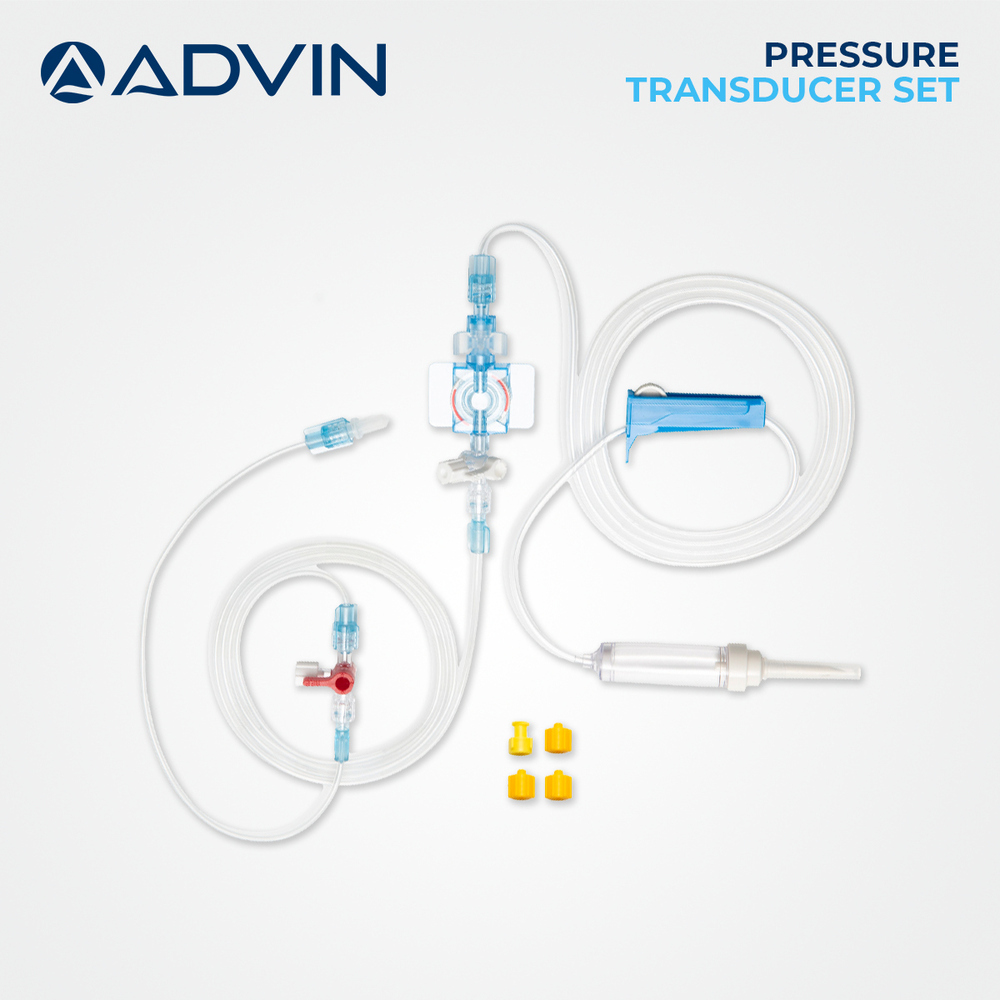



Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Cardiology Products Category
Cardiac Wrist Compression Device
Price 5.5 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Real-Time Operation : Yes
Accuracy : 3 mmHg
Dimension (L*W*H) : 17 x 7 x 5 cm
Use : Clinical, Hospital, Personal Care
Clot Retrieval Catheter
Price 210 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Real-Time Operation : Yes
Accuracy : High retrieval efficiency (as per clinical data)
Dimension (L*W*H) : Length: 150 cm (typical), Outer Diameter: 6F
Use : Neurovascular and Peripheral Embolism Clot Retrieval

 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry


 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese