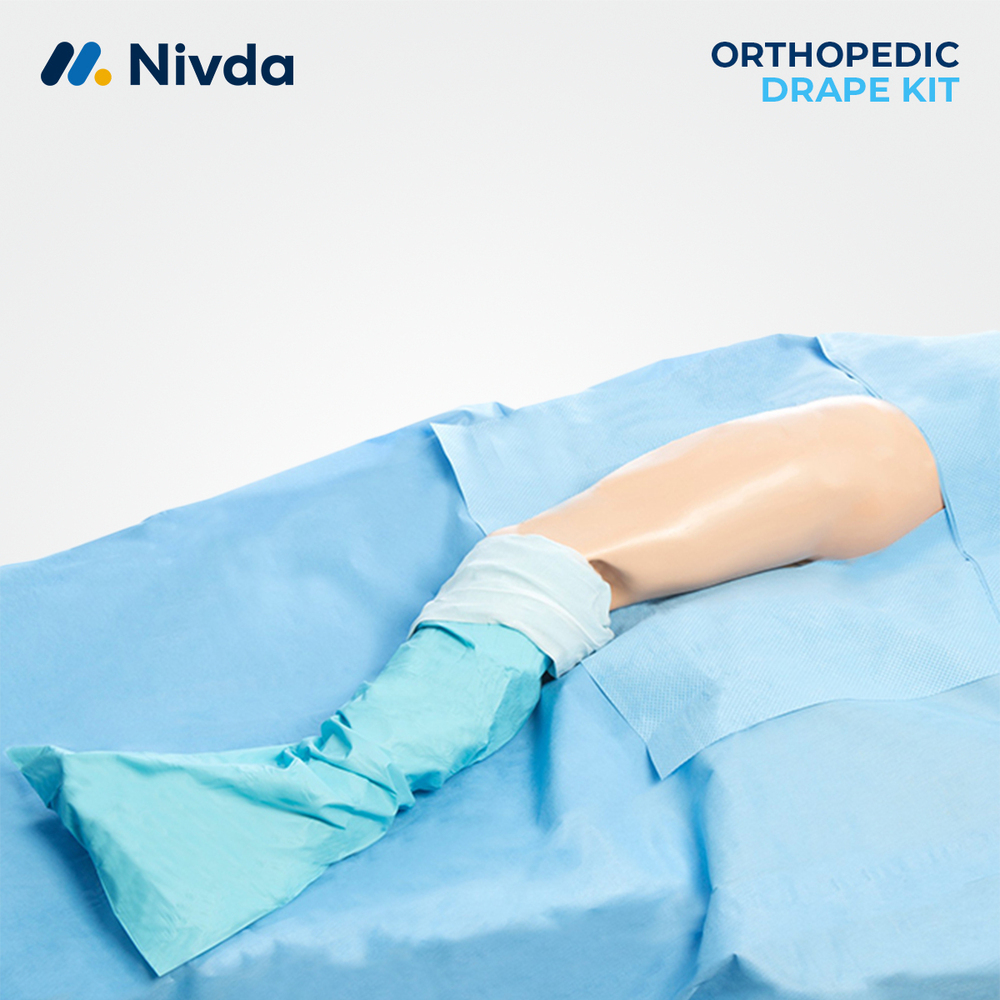
Surgical Site Isolation Drape
Price 5.10 USD ($)/ Piece
Surgical Site Isolation Drape Specification
- Function
- Site isolation and field barrier during surgical procedures
- Usage Type
- Hospital, Clinic, Operating Room
- Storage Instructions
- Store in a cool, dry place away from direct sunlight
- Shelf Life
- 5 years (when stored properly)
- Instruments Type
- Surgical Drape
- Features
- Adhesive area for secure placement, Fluid repellent, Tear resistant, Sterile packaging, Lint free
- Equipment Type
- Surgical Site Isolation Drape
- Material
- Medical Grade SMS Non-Woven Fabric
- Condition
- New
- Technology
- Sterile Barrier Technology
- Portable
- Yes
- Wall Mounted
- No
- Operating Type
- Disposable - Single Use
- Use
- Covering and isolating the surgical site to prevent infection
- Dimension (L*W*H)
- Available in multiple standard sizes (e.g., 75cm x 90cm)
- Weight
- Lightweight (variable per size)
- Color
- Blue
- Sterility
- Sterile, Ethylene Oxide (ETO) sterilized
- Application
- Reduces risk of surgical site infections (SSIs)
- Pack Size
- Individually packed in sterile wraps
- Disposability
- 100% disposable after single use
- Adhesive Strip
- Yes, integrated medical-grade adhesive for fixation
- Permeability
- Breathable yet impervious to fluids
- Shape
- Rectangular with fenestration (window) on request
- Latex Content
- Latex free
About Surgical Site Isolation Drape
Nivda Drapes is globally leading manufacturer and supplier of Surgical Site Isolation Drape. Surgical Site Isolation Drapes are designed to create a protective sterile barrier around the operative area during surgical procedures. They help separate the surgical site from surrounding surfaces, reducing the risk of contamination in the operating environment. Nivda Drapes provides reliable disposable drapes manufactured to support infection prevention and maintain sterile surgical conditions. With quality materials and sterile processing standards, these drapes assist healthcare professionals in maintaining a safe and organized surgical field throughout the procedure.Advanced Features:
Sterile disposable drapes designed for surgical site isolation
Helps maintain a clean and controlled operative environment
Fluid-resistant material for effective barrier protection
Easy placement around the surgical site
Single-use design to support hygiene and infection control
Surgical Site Isolation Drape, Isolation Surgical Drape, Sterile Procedure Drape, Disposable Surgical Drape, Medical Isolation Drape, OT Surgical Drape, Non Woven Surgical Drape
غطاء عزل موقع الجراØØ©ØŒ غطاء عزل جراØÙŠØŒ غطاء إجراءات معقمة، غطاء جراØÙŠ للاستخدام مرة واØدة، غطاء عزل طبي، غطاء جراØÙŠ لغرÙØ© العمليات، غطاء جراØÙŠ غير منسوج
ХирургичеÑÐºÐ°Ñ Ð¸Ð·Ð¾Ð»ÑÑ†Ð¸Ð¾Ð½Ð½Ð°Ñ Ð¿Ñ€Ð¾ÑÑ‚Ñ‹Ð½Ñ Ð´Ð»Ñ Ð¾Ð¿ÐµÑ€Ð°Ñ†Ð¸Ð¾Ð½Ð½Ð¾Ð³Ð¾ полÑ, изолÑÑ†Ð¸Ð¾Ð½Ð½Ð°Ñ Ñ…Ð¸Ñ€ÑƒÑ€Ð³Ð¸Ñ‡ÐµÑÐºÐ°Ñ Ð¿Ñ€Ð¾ÑтынÑ, ÑÑ‚ÐµÑ€Ð¸Ð»ÑŒÐ½Ð°Ñ Ð¿Ñ€Ð¾Ñ†ÐµÐ´ÑƒÑ€Ð½Ð°Ñ Ð¿Ñ€Ð¾ÑтынÑ, Ð¾Ð´Ð½Ð¾Ñ€Ð°Ð·Ð¾Ð²Ð°Ñ Ñ…Ð¸Ñ€ÑƒÑ€Ð³Ð¸Ñ‡ÐµÑÐºÐ°Ñ Ð¿Ñ€Ð¾ÑтынÑ, медицинÑÐºÐ°Ñ Ð¸Ð·Ð¾Ð»ÑÑ†Ð¸Ð¾Ð½Ð½Ð°Ñ Ð¿Ñ€Ð¾ÑтынÑ, хирургичеÑÐºÐ°Ñ Ð¿Ñ€Ð¾ÑÑ‚Ñ‹Ð½Ñ Ð´Ð»Ñ Ð¾Ð¿ÐµÑ€Ð°Ñ†Ð¸Ð¾Ð½Ð½Ð¾Ð¹, Ð½ÐµÑ‚ÐºÐ°Ð½Ð°Ñ Ñ…Ð¸Ñ€ÑƒÑ€Ð³Ð¸Ñ‡ÐµÑÐºÐ°Ñ Ð¿Ñ€Ð¾ÑтынÑ
Campo quirúrgico de aislamiento, campo quirúrgico de aislamiento, campo estéril para procedimientos, campo quirúrgico desechable, campo de aislamiento médico, campo quirúrgico para quirófano, campo quirúrgico no tejido
Champ opératoire d'isolement, Champ opératoire stérile, Champ opératoire à usage unique, Champ d'isolement médical, Champ opératoire non tissé
Advanced Sterile Barrier Technology
Engineered with cutting-edge Sterile Barrier Technology, our isolation drape offers exceptional protection by combining breathability with fluid impermeability. Sterile packaging and ETO sterilization ensure the highest standards of patient safety and infection control. The lightweight, lint-free construction also minimizes contamination and is comfortable for both practitioners and patients.
Secure Fit with Medical-Grade Adhesive
Featuring a specialized, integrated medical-grade adhesive strip, the Surgical Site Isolation Drape adheres firmly to the skin, maintaining placement throughout lengthy procedures. The user-friendly design accommodates quick and precise positioning, while the adhesive ensures the drape does not shift, reducing the risk of accidental exposure or contamination.
Convenient and Versatile Design
Designed for ease of use across hospitals, clinics, and operating rooms, this drape comes in multiple sizes and is available with fenestration on request for varied surgical needs. The 100% disposable and latex-free construction promotes infection control and reduces allergen risks. Individually wrapped for single-use convenience, it maintains sterility right up to the point of application.
FAQ's of Surgical Site Isolation Drape:
Q: How does the Surgical Site Isolation Drape help reduce surgical site infections (SSIs)?
A: The drape provides a sterile, fluid-impervious barrier between the surgical site and the external environment. Its advanced SMS non-woven fabric restricts microbial transmission, while ETO sterilization and lint-free design further decrease contamination risks, helping to significantly lower the incidence of SSIs.Q: What is the process for applying this surgical drape during a procedure?
A: First, the sterile drape is removed from its individual packaging. It is then positioned over the surgical area, with the integrated medical-grade adhesive strip pressed gently onto the patient's skin for secure fixation. If a fenestration (window) is required, the drape is aligned accordingly to provide access while maintaining a sterile field.Q: When should the drape be used and disposed of?
A: The drape is designed for single-use during a single surgical procedure. It should be applied before surgery begins and disposed of immediately after the procedure in accordance with standard hospital protocols for biohazardous waste to maintain infection control.Q: Where can this sterile isolation drape be utilized?
A: This drape is ideal for use in operating rooms, hospitals, and clinical settings where sterile conditions are critical. Its versatility and range of available sizes make it suitable for various surgical operations and patient types.Q: What are the main benefits of using a drape with integrated adhesive and sterile barrier technology?
A: The built-in medical-grade adhesive ensures the drape remains securely in place, preventing unwanted shifts or exposure. Sterile barrier technology provides effective infection control, fluid repellency, and a breathable, tear-resistant shield, protecting both patients and surgical staff.Q: What storage conditions are recommended for maintaining product sterility?
A: For optimal shelf life and sterility, store the drape in a cool, dry place away from direct sunlight. Following these storage guidelines ensures it remains safe and effective for up to five years from the date of manufacture.Q: Is this surgical drape latex-free, and how does that benefit patients?
A: Yes, the drape is 100% latex-free. This eliminates the risk of allergic reactions in patients and clinicians sensitive to latex, making it a safer choice for all surgical environments.






Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Disposable Drapes Category
Gynecology Drape
Price 4.10 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Function : Provides sterile barrier during gynecological procedures
Usage Type : Single Use
Technology : Ultrasonic Sealing
Storage Instructions : Store in a cool, dry place away from direct sunlight
Nivda Waterproof Orthopedic Drapes
Price 6 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Function : Fluid barrier, Protect surgical site
Usage Type : Single Use (Disposable)
Technology : Advanced Layered Barrier
Storage Instructions : Keep in a cool, dry place; avoid direct sunlight
Shoulder U Drape Kit
Price 8.50 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Function : Creates a sterile field during shoulder surgery
Usage Type : Singleuse
Technology : Ultrasonic Sealing
Storage Instructions : Store in a cool, dry place, away from direct sunlight
Disposable Gown
Price 2.56 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Function : Personal Protective Apparel
Usage Type : Disposable
Technology : Spunbond / SMS
Storage Instructions : Store in a cool, dry place away from direct sunlight

 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry



 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese