Angiography Guide Wires
Price 15 USD ($)/ Piece
Angiography Guide Wires Specification
- Instruments Type
- Interventional Cardiovascular Instrument
- Features
- Flexible, smooth navigation, kink resistance, radiopaque tip
- Function
- Assisting catheter placement in vessels
- Shelf Life
- 5 Years
- Accuracy
- High precision navigation
- Storage Instructions
- Store in a cool, dry place, away from direct sunlight
- Usage Type
- Single Use (Disposable)
- Equipment Type
- Angiography Guide Wires
- Material
- Stainless Steel
- Condition
- New
- Technology
- Advanced Hydrophilic Coating
- Portable
- Yes
- Wall Mounted
- No
- Real-Time Operation
- Yes
- Noise Level
- Silent
- Operating Type
- Manual
- Use
- For guiding catheters during angiography procedures
- Power Source
- Manual
- Dimension (L*W*H)
- 150 cm (length standard)
- Weight
- Lightweight
- Color
- Silver or Blue Coating
- Disposability
- Meant for single procedure use
- Manufacturer Standard
- Meets ISO and CE standards
- Packaging
- Individually packed in sterile pouch
- Tip Material
- Soft Flexible Polymer
- Radiopacity
- Radiopaque tip for visibility under fluoroscopy
- Diameter
- 0.018 - 0.038 inches (various sizes available)
- Compatibility
- Compatible with standard angiographic catheters
- Sterility
- Sterile, EO (Ethylene Oxide) Sterilized
- Resistant Feature
- Corrosion and kink resistant
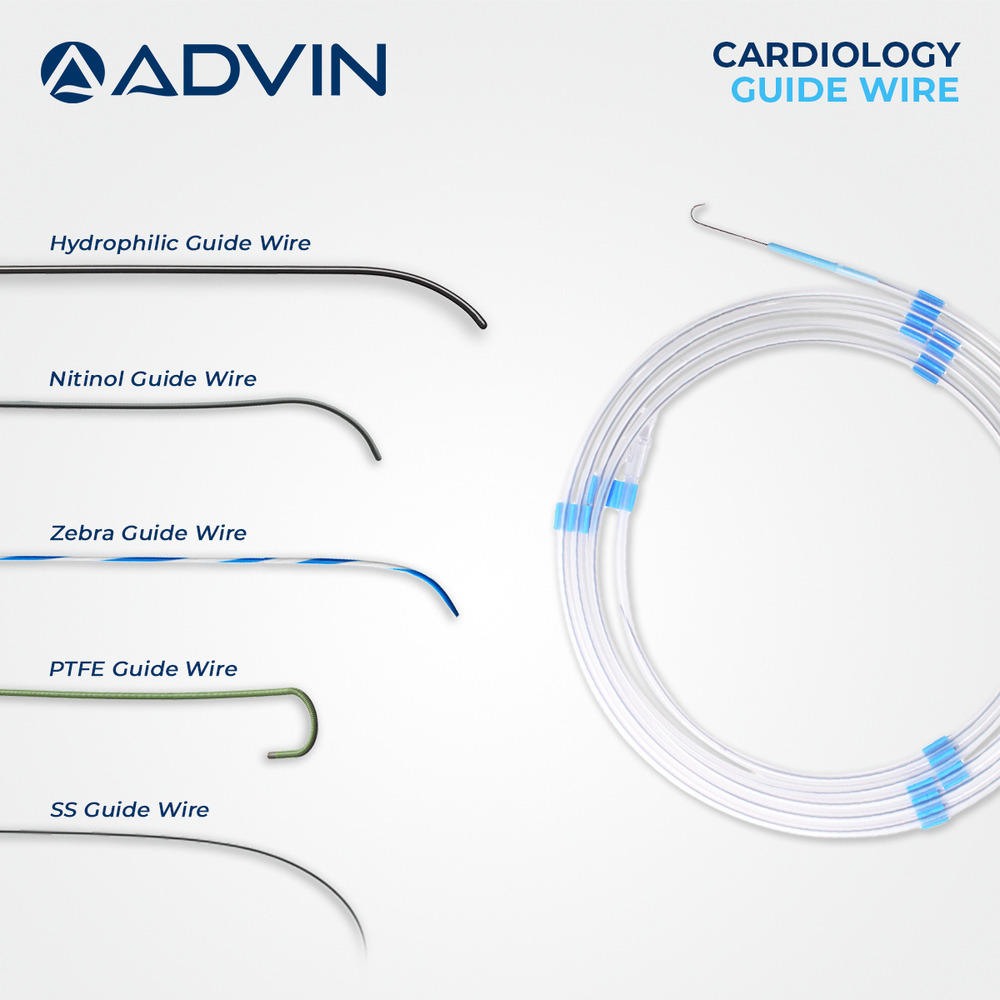
- Coating Type
- Hydrophilic or PTFE Coated
- Tip Shape
- Straight or Angled Tip
About Angiography Guide Wires
Advin Health Care is a trusted manufacturer of Angiography Guide Wires designed to meet international quality standards. These guide wires are widely used in angiographic and diagnostic procedures to enable smooth vascular access and precise catheter placement, ensuring reliable performance during imaging and vascular assessment.
Key Features
- Smooth surface for easy vascular navigation
- Excellent torque control for precise positioning
- High flexibility for navigating complex vascular anatomy
- Atraumatic tip design for enhanced patient safety
Specifications
- Type: Angiography Guide Wire
- Surface: Standard / PTFE-coated / Hydrophilic options available
- Usage: Single-use / Sterile
- Application: Angiography and diagnostic vascular procedures
Certifications
CE, ISO 13485, and ISO 9001 certified
Product Keywords
Angiography Guide Wire, Diagnostic Guide Wire, Vascular Guide Wire, Medical Guide Wire, Interventional Guide Wire
, , , ,
Gua de angiografa, gua de diagnstico, gua vascular, gua mdica, gua intervencionista
Fil guide d'angiographie, fil guide de diagnostic, fil guide vasculaire, fil guide mdical, fil guide interventionnel
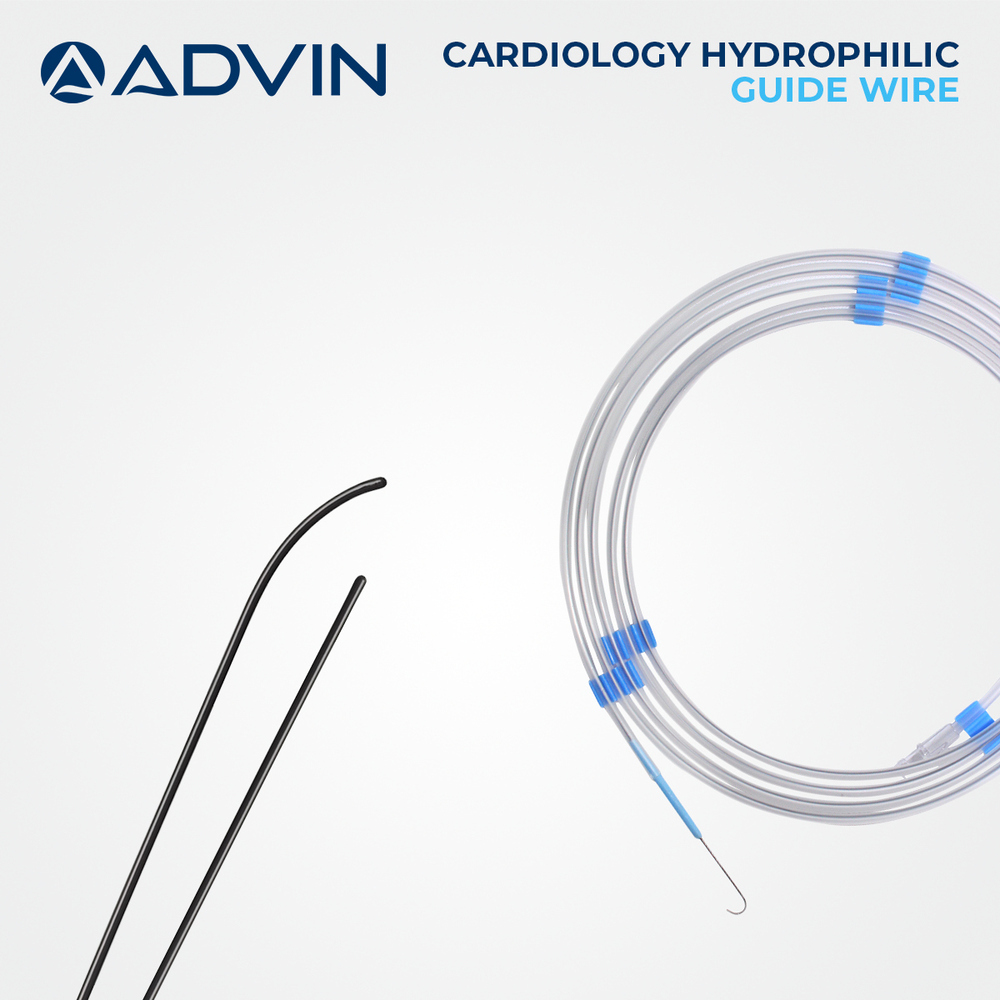
Advanced Hydrophilic and PTFE Coatings
Our angiography guide wires utilize cutting-edge hydrophilic or PTFE coatings that significantly enhance smoothness and ease of navigation through vascular pathways. This advanced coating minimizes friction, allowing precise and gentle catheter placement while reducing the risk of vessel trauma during procedures.
Sterile, Single-Use Safety
Each guide wire is EO (Ethylene Oxide) sterilized and individually packed, ensuring complete sterility before use. Designed strictly for single-procedure use, they help prevent cross-contamination and infection while maintaining the highest patient safety standards.
Radiopaque Tip for Enhanced Visibility
Featuring a soft, flexible radiopaque polymer tip, these guide wires provide clear visibility under fluoroscopy, assisting clinicians in accurate navigation and placement. The tip is available in both straight and angled shapes to suit various procedural needs and patient anatomies.
FAQ's of Angiography Guide Wires:
Q: How are these angiography guide wires sterilized before use?
A: The angiography guide wires are sterilized using Ethylene Oxide (EO) and are individually packed in sterile pouches to maintain their sterility until opened for use in procedures.Q: What tip shapes and materials are available for these guide wires?
A: These guide wires are available with either straight or angled tips, both made from a soft, flexible polymer. This ensures gentle navigation and adaptation to different vessel anatomies during angiographic procedures.Q: When should these guide wires be used in angiography?
A: These guide wires are intended for single-use during catheter placement in vessels, primarily during angiography procedures to assist with accurate, safe, and smooth navigation within the vascular system.Q: Where can these guide wires be utilized?
A: The guide wires can be utilized in hospitals, clinics, and interventional cardiovascular facilities where angiographic procedures or catheter placements are performed, meeting global standards for sterility and safety.Q: What is the process for using the angiography guide wire?
A: The wire is manually inserted into the vascular system via an introducer sheath, then advanced under fluoroscopic guidance. The hydrophilic/PTFE coating and flexible tip ensure smooth movement and precise positioning for catheter guidance.Q: What are the primary benefits of using these guide wires?
A: Key benefits include high precision navigation, smooth catheter placement due to advanced coating, kink and corrosion resistance, radiopaque tip visibility, and strict sterilization-ensuring safety and efficacy in every procedure.Q: How should the guide wires be stored before use?
A: For optimal longevity and sterility, store the guide wires in a cool, dry place, away from direct sunlight, following their five-year shelf life guidelines.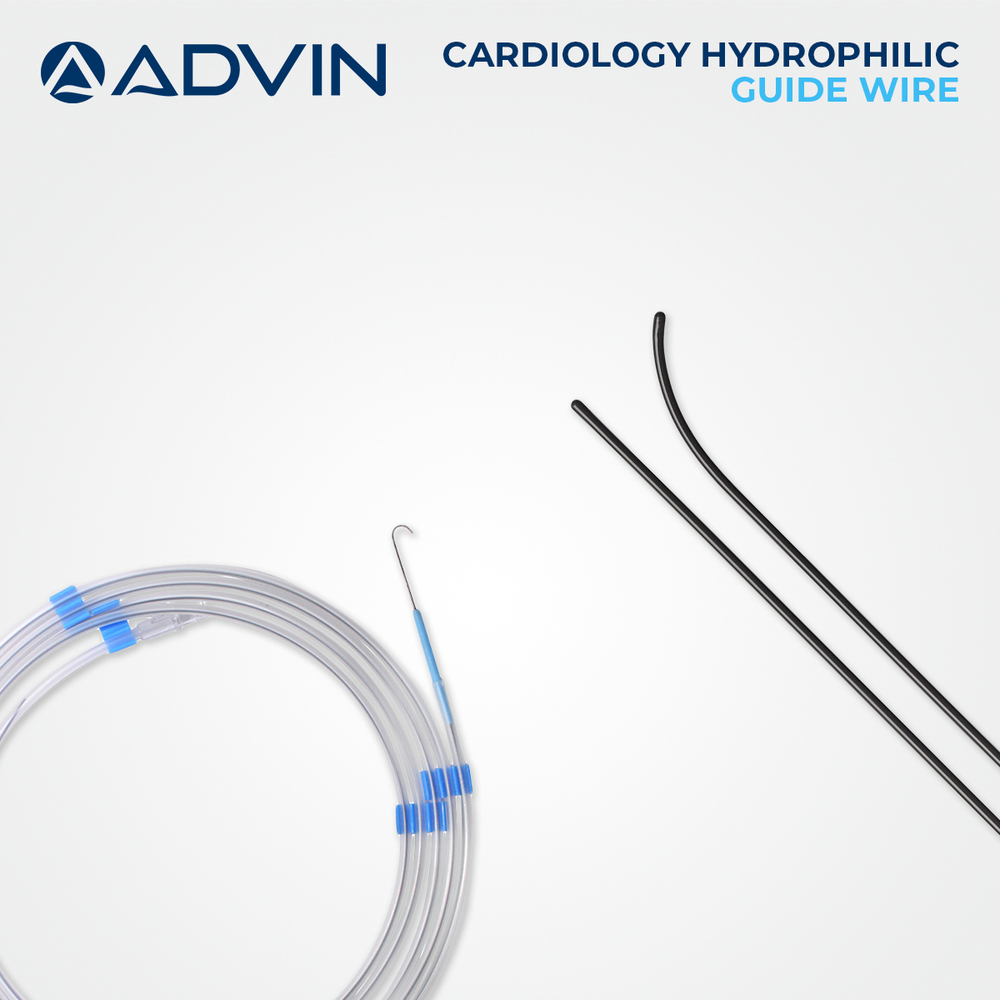



Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Cardiology Products Category
Cardiovascular Disposable Products Archives
Price 50 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Use : Cardiovascular Procedures
Condition : New
Operating Type : Manual
Material : Other, Medical Grade PVC, LatexFree Polymers
Two Way Manifolds
Price 2 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Use : Hospital
Condition : New
Operating Type : Manual
Material : Plastic

 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry



 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese