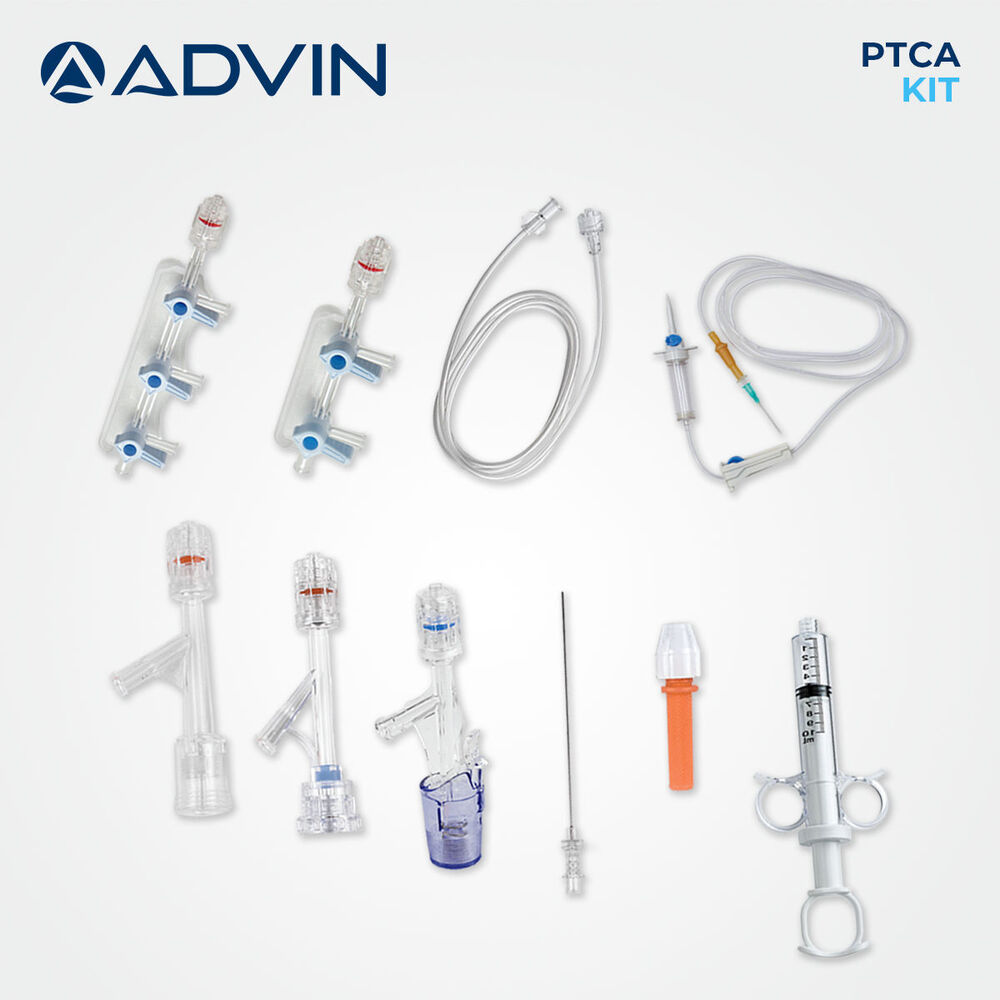
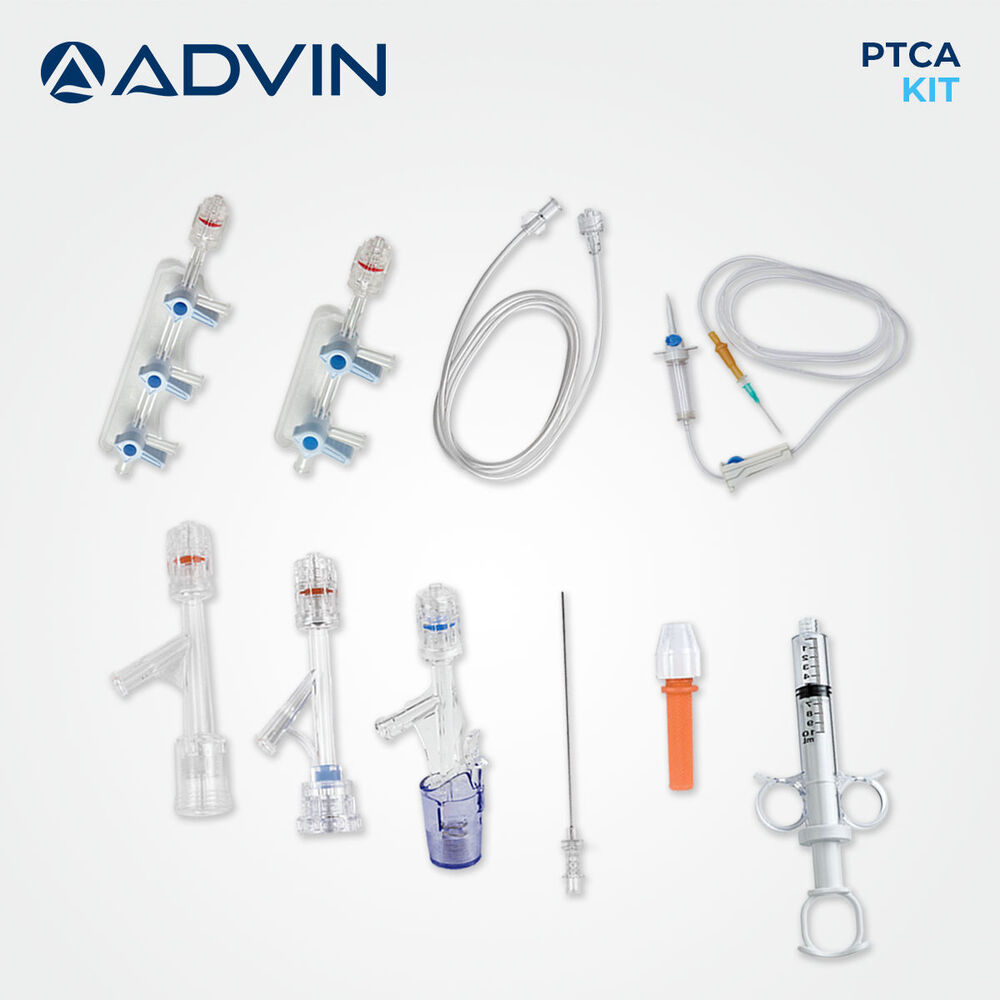
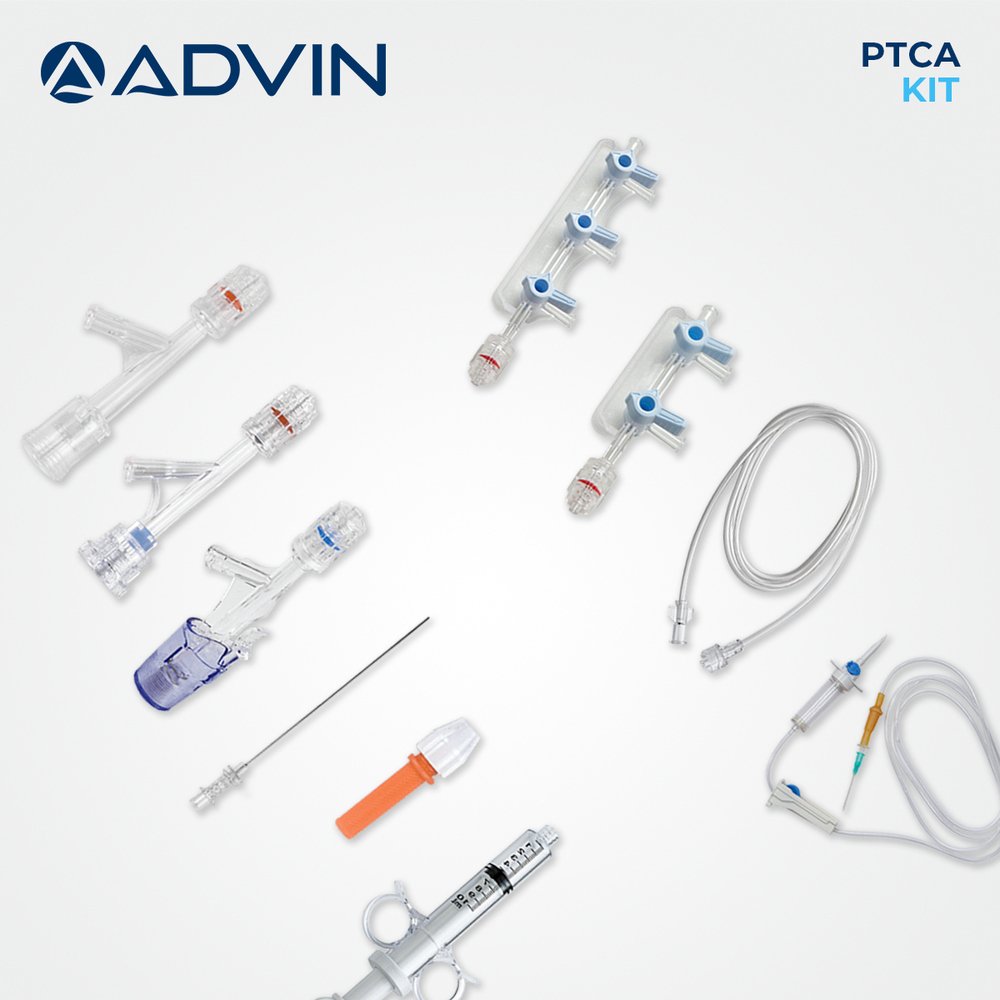
Coronary Angioplasty Kit
Price 18 USD ($)/ Piece
Coronary Angioplasty Kit Specification
- Instruments Type
- Interventional cardiology kit
- Measurement Range
- Catheter sizes per requirements
- Function
- Facilitates dilation of narrowed coronary arteries
- Features
- Comprehensive kit, ready-to-use, disposable
- Usage Type
- Hospital, cardiac catheterization lab
- Shelf Life
- 3 years
- Accuracy
- High precision for controlled delivery
- Storage Instructions
- Store in a cool, dry place, away from sunlight
- Equipment Type
- Coronary Angioplasty Kit
- Material
- Medical grade polymer and stainless steel
- Condition
- Sterile, Single-use
- Technology
- Minimally invasive intervention
- Portable
- Yes
- Wall Mounted
- No
- Real-Time Operation
- Yes
- Noise Level
- Silent
- Operating Type
- Manual
- Use
- Angioplasty procedure for coronary artery disease
- Dimension (L*W*H)
- Customized as per kit contents
- Weight
- Lightweight, typically < 1 kg
- Color
- Translucent/White (kit components)
- Balloon Catheter
- PTCA Balloon catheter, various diameters
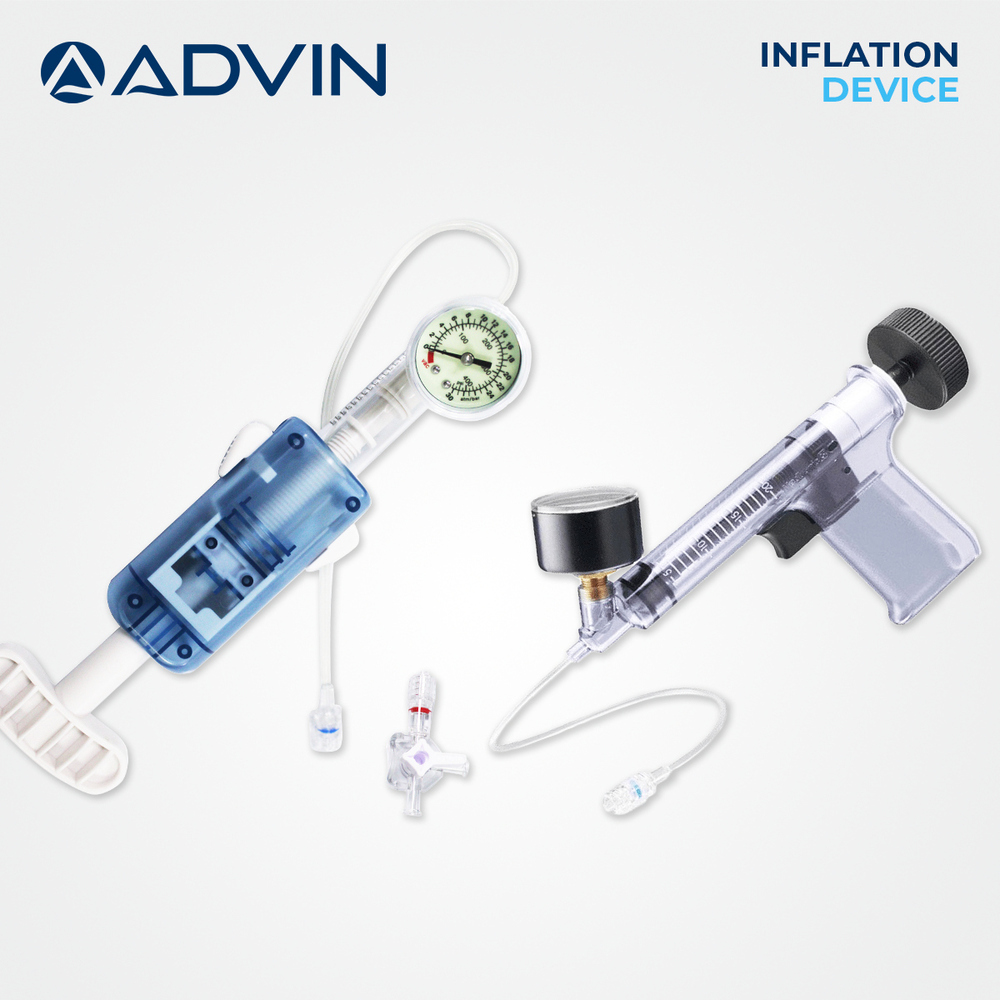
- Syringe
- Included for inflation/deflation
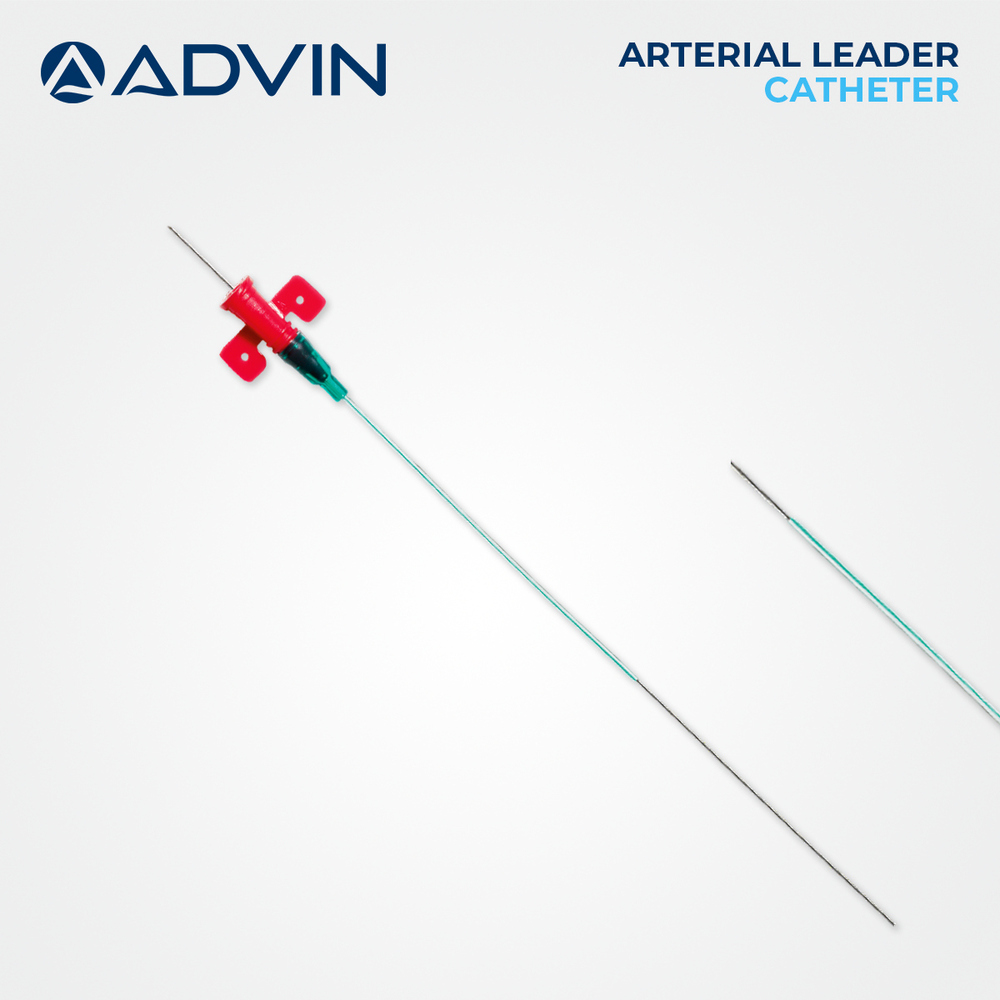
- Catheter Guidewire
- Included, 0.014'' or 0.018'' compatible
- Dilator
- Yes, included in kit
- Compatibility
- Compatible with standard angioplasty equipment
- Extension Tube
- Yes, for ease of access
- Introducer Sheath
- Standard sizes included
- Radiopaque Marking
- Available for precise placement
- Sterility Assurance
- ETO (Ethylene Oxide) sterilized
- Intended Patient Population
- Adults and pediatric patients (as specified)
- User Instruction
- Provided with kit
- Latex Free
- Yes
- Regulatory Compliance
- CE Marked, ISO 13485 certified
- Packaging
- Sealed, tamper-proof packaging
About Coronary Angioplasty Kit
The coronary angioplasty kit is designed to support smooth and efficient coronary interventions by providing carefully selected procedural components. Its use helps clinicians maintain consistency and control throughout angioplasty procedures. Advin Health Care ensures the kit meets clinical expectations for reliability and procedural readiness. The kit supports streamlined workflow in cath labs and contributes to effective patient treatment during coronary interventions.
Advanced FeaturesComplete procedural kit for coronary angioplasty Designed to support controlled and reliable vessel dilation Compatible with standard coronary intervention devices Intended for single-use clinical applications
Certifications
We are certified with CE, ISO 13485, and ISO 9001 standards.
Product Keywords
Coronary Angioplasty Kit, PTCA Kit, Percutaneous Coronary Intervention Kit, Interventional Cardiology Kit, Coronary Balloon Angioplasty Kit, Single-Use Angioplasty Kit
UUU1 UU1U UaU UUU1 UU1U UaU 1U UU UU UUU1 Ua U UaU 1U UU UU UUU1 UUU Ua UU UUU1 UU1U UaU U UUU UUU1 UU1U UaU UU3a U U U
34N N o34N3412N12341 123 34NN o , 1234N N , 1234N N NNNo3412341 o34N3412N12341 12NN212N , 1234N N 12NN212N 341212341 oN 34343 , 1234N N o34N3412N12341 341212341 123 34NN o , 34 1234N342N1 1234N N 123 34NN o .
Kit de angioplastia coronaria, kit de PTCA, kit de intervenciA3n coronaria percutAnea, kit de cardiologAa intervencionista, kit de angioplastia coronaria con balA3n, kit de angioplastia de un solo uso
Kit d'angioplastie coronaire, kit d'angioplastie coronaire percutanAe (PTCA), kit d'intervention coronaire percutanAe, kit de cardiologie interventionnelle, kit d'angioplastie par ballonnet coronaire, kit d'angioplastie A usage unique
Comprehensive Coronary Angioplasty Solution
The Coronary Angioplasty Kit is designed as an all-in-one solution, providing every vital instrument for percutaneous coronary intervention. With components like guidewire, balloon catheter with various diameters, introducer sheath, and extension tubing, clinicians benefit from convenience and precise operative control. The kit's ergonomic design and compatibility with standard angioplasty equipment make it an optimal choice for hospitals and cardiac cath labs.
Precision and Safety Assured
Radiopaque markings on the balloon catheter ensure accurate visualization and placement during procedures. The kit is ETO sterilized and latex-free, which minimizes the risk of infection and allergic reaction. Its CE Mark and ISO 13485 certification guarantee conformity to international safety and quality standards, while sealed, tamper-proof packaging preserves sterility up to its three-year shelf life.
FAQ's of Coronary Angioplasty Kit:
Q: How is the Coronary Angioplasty Kit used during procedures?
A: The Coronary Angioplasty Kit is used for minimally invasive dilation of narrowed coronary arteries. After vascular access is obtained, the introducer sheath and dilator facilitate entry, the guidewire is advanced to the target lesion, and the PTCA balloon catheter is maneuvered over the wire. The included syringe allows for controlled inflation and deflation of the balloon, guided by radiopaque markers for accurate placement.Q: What are the benefits of choosing this angioplasty kit?
A: This kit streamlines the procedure with all necessary components ready for use, optimizing workflow in cardiac interventions. It ensures high precision with radiopaque markings and offers enhanced safety through ETO sterilization, latex-free materials, and regulatory compliance. Its comprehensive design minimizes preparation time and supports better procedural outcomes.Q: When should this coronary angioplasty kit be used?
A: The kit is intended for use during elective or emergency angioplasty procedures in adult or pediatric patients, as specified, who present with coronary artery stenosis and require percutaneous transluminal coronary angioplasty (PTCA) to restore blood flow.Q: Where can this angioplasty kit be utilized?
A: It is suitable for use in cardiac catheterization laboratories and hospital settings equipped for interventional cardiology procedures. Its portability and comprehensive contents make it ideal for both scheduled and emergent interventions.Q: What instructions are provided for proper usage?
A: Each kit includes detailed user instructions outlining preparation, optimal usage, inflation/deflation technique, and proper handling of each component to ensure clinical safety and procedural accuracy.Q: How does the kit ensure patient and operator safety?
A: Sterility is maintained through ETO sterilization and tamper-proof packaging. The latex-free construction reduces the risk of allergic reactions, while radiopaque markers assist in safe, precise placement. Compliance with CE Mark and ISO 13485 standards upholds product reliability and safety.Q: What storage and shelf life requirements are associated with the kit?
A: The kit should be stored in a cool, dry location away from direct sunlight to preserve sterility. It has a shelf life of three years when unopened and stored under recommended conditions.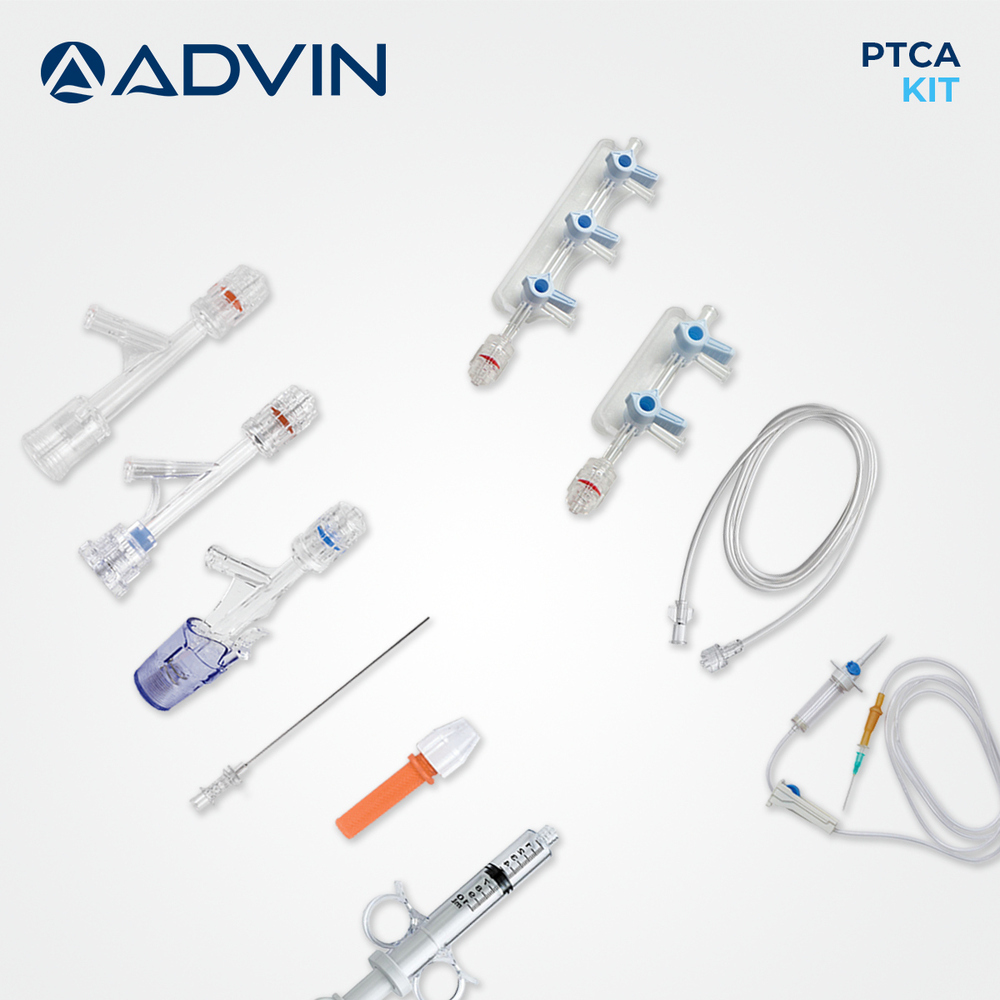




Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Cardiology Products Category
Temporary Pacemaker Leads
Price 30 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Equipment Type : Temporary Pacemaker Leads
Material : Other, PTFEcoated stainless steel
Operating Type : Other, Manual insertion
Accuracy : High conduction accuracy
Single-Use PTCA Kit
Price 18 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Equipment Type : SingleUse PTCA Kit
Material : Other, Medical Grade Polymer, Stainless Steel
Operating Type : Manual
Accuracy : High Precision Components

 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese