Single-Use PTCA Kit
Price 18 USD ($)/ Piece
Single-Use PTCA Kit Specification
- Accuracy
- High Precision Components
- Storage Instructions
- Store in a cool, dry place, away from sunlight
- Shelf Life
- 3 Years
- Usage Type
- Single Use
- Function
- Facilitate Balloon Angioplasty procedures
- Features
- Sterile, Single-Use, Ready-to-use, CE Marked
- Instruments Type
- Cardiovascular Intervention Kit
- Equipment Type
- Single-Use PTCA Kit
- Material
- Medical Grade Polymer, Stainless Steel
- Condition
- New
- Technology
- Disposable, Sterile Kit
- Portable
- Yes
- Wall Mounted
- No
- Real-Time Operation
- No
- Noise Level
- Silent Operation
- Operating Type
- Manual
- Use
- Percutaneous Transluminal Coronary Angioplasty
- Dimension (L*W*H)
- Custom for Individual Components
- Weight
- Lightweight, Approx. 200 g per kit
- Color
- Transparent / Clear
- Packaging Type
- Blister Pack
- Sterilization Method
- Ethylene Oxide (ETO) Sterile
- Intended User
- Cardiologists, Interventional Cardiologists, Cath Lab Professionals
- Expiry Date Marked
- Yes
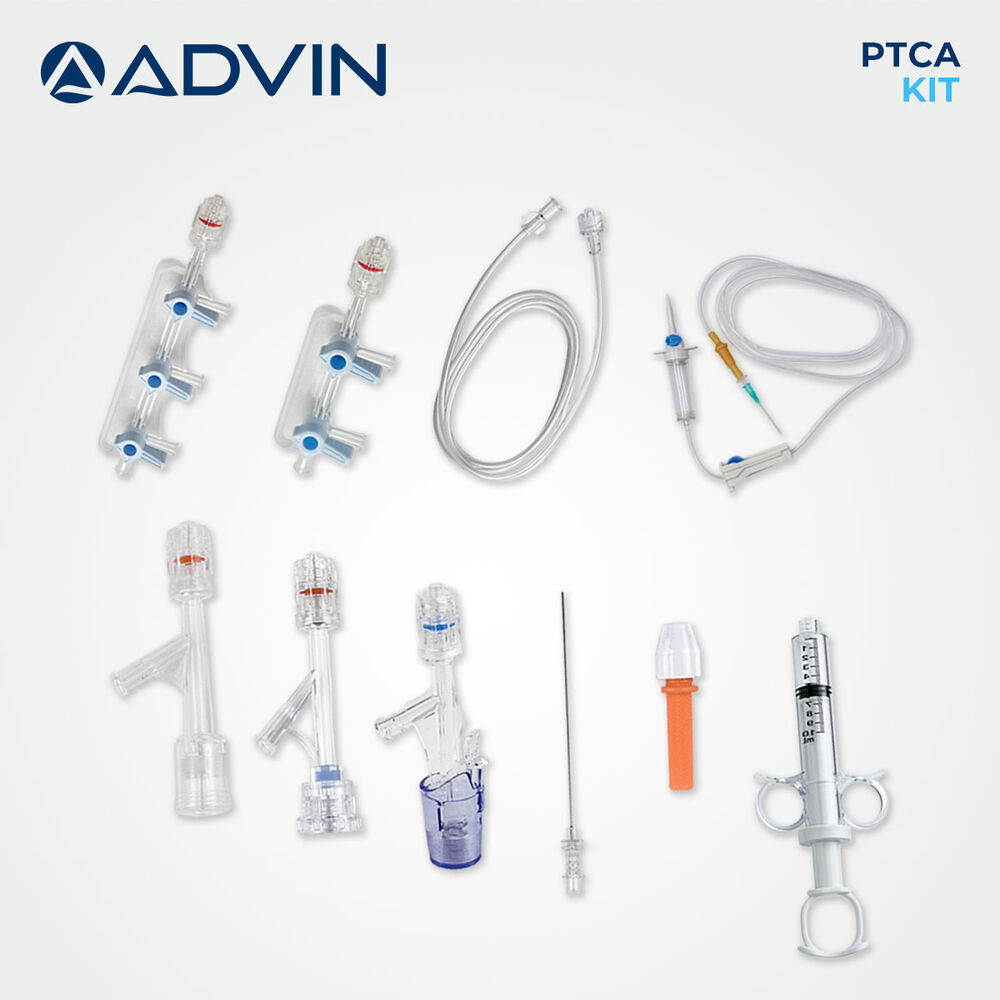
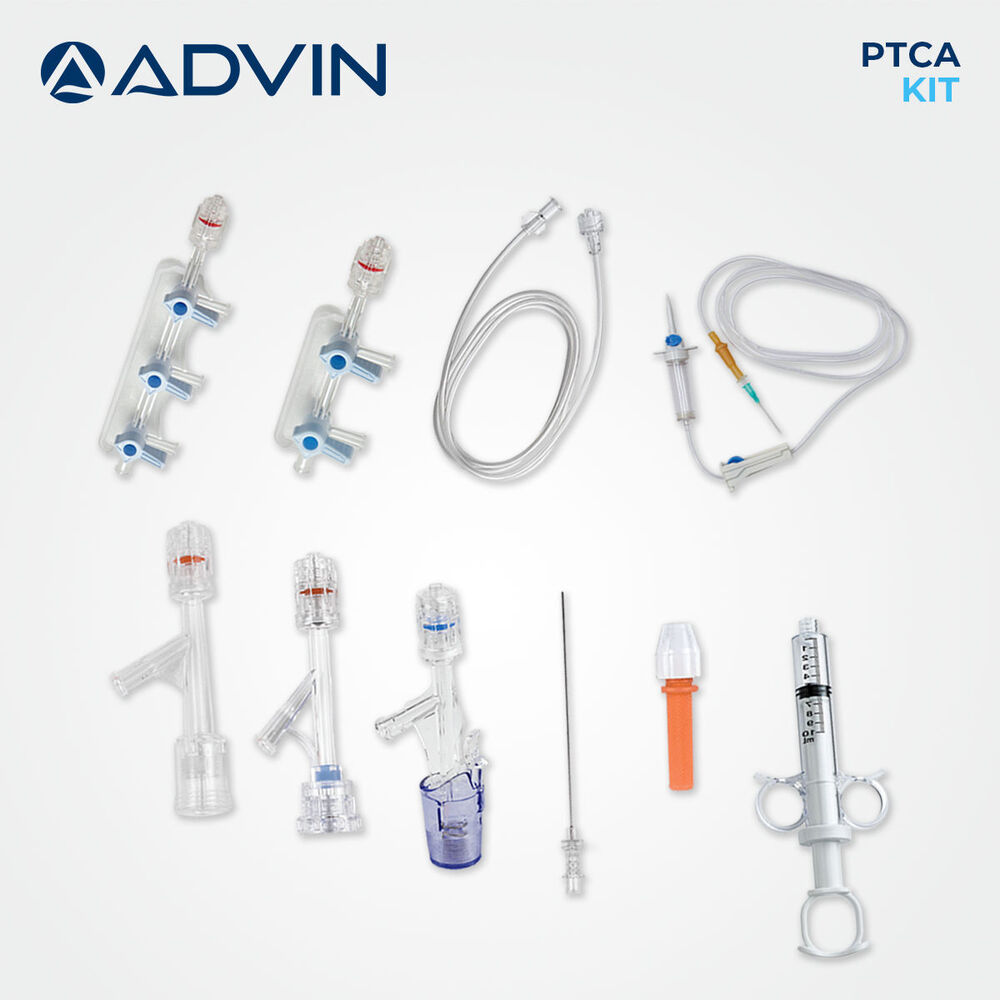
- Kit Contents
- Introducer Needle, Guidewire, Y Connector, PTCA Catheter, Syringe, Stopcock, Extension Tube
- Radiopacity
- Radiopaque Markers Included
- Compliance Standard
- ISO 13485, CE Certified
- Latex Content
- Latex-Free
- Temperature for Storage
- 15C to 30C (59F to 86F)
- Disposability
- Single Patient Use Only
- Application Site
- Coronary Arteries
About Single-Use PTCA Kit
Advin Health Care is a trusted manufacturer of Single-Use PTCA Kits designed to meet international quality standards. These kits are used in percutaneous coronary interventions for the safe and effective treatment of coronary artery stenosis.
Key Features
- Complete kit for percutaneous transluminal coronary angioplasty (PTCA)
- Includes guide wire, balloon catheter, and Y-connector for streamlined procedures
- Sterile, single-use components to ensure patient safety
- High-quality materials for optimal performance and reliability
- Compatible with standard angioplasty equipment and monitoring systems
Certifications
We are certified with CE, ISO 13485, and ISO 9001 standards.
Product Keywords
Single-Use PTCA Kit, PTCA Kit, Percutaneous Coronary Intervention Kit, Coronary Angioplasty Kit, Single-Use Angioplasty Kit, Interventional Cardiology Kit
UU UU1U UaU 1U UU UU UU3a U UU U UU UU1U UaU 1U UU UU UU Ua U UaU 1U UU UU UU UU1U UaU UU UU1U UaU UU3a U UU U UU UUU Ua UU
34N N 34 1234N342341 NNNo3412341 o34N3412N12341 123 34NN o (), 1234N N , 1234N N NNNo3412341 o34N3412N12341 12NN212N , 1234N N o34N3412N12341 123 34NN o , 1234N N 34 1234N342341 123 34NN o , 1234N N 12NN212N 341212341 oN 34343 .
Kit de PTCA de un solo uso, Kit de PTCA, Kit de intervenciA3n coronaria percutAnea, Kit de angioplastia coronaria, Kit de angioplastia de un solo uso, Kit de cardiologAa intervencionista
Kit d'angioplastie coronaire percutanAe A usage unique, kit d'angioplastie coronaire, kit d'angioplastie A usage unique, kit de cardiologie interventionnelle
Comprehensive PTCA Kit for Cardiac Intervention
This single-use PTCA kit provides all essential components required for safe and effective percutaneous transluminal coronary angioplasty. Designed for quick setup and ease of use, it helps streamline cath lab workflows and is compatible with radiopaque imaging technologies. Each item has been carefully selected and assembled using medical-grade materials for optimum precision and patient safety.
Sterilized and Safety Assured
The kit is sterilized using ethylene oxide (ETO), ensuring maximum sterility for every component. Packaged in a latex-free blister pack, it minimizes the risk of allergic reactions and is CE certified, meeting global safety and quality standards for cardiovascular intervention kits.
Ready-to-Use and Single-Patient Focused
With its single-use format, the PTCA kit reduces chances of cross-contamination while offering unparalleled convenience to professionals. Each component is pre-assembled and intended for immediate use, helping cath lab teams maintain efficiency and hygiene during coronary procedures.
FAQ's of Single-Use PTCA Kit:
Q: How should the Single-Use PTCA Kit be stored to maintain sterility?
A: The kit should be stored in a cool, dry place away from direct sunlight, with ambient temperatures maintained between 15C and 30C (59F to 86F) to ensure product integrity and sterility until use.Q: What are the individual components included in each PTCA kit?
A: Each kit contains an introducer needle, guidewire, Y connector, PTCA catheter, syringe, stopcock, and extension tube, all pre-assembled and ready for immediate use in angioplasty procedures.Q: When can the Single-Use PTCA Kit safely be used following shipment?
A: The kit is sterile upon arrival and ready to use immediately if the packaging is intact and it is before the marked expiry date. Always inspect for package integrity prior to opening.Q: Where is the PTCA Kit primarily used and who are the intended users?
A: This kit is designed for application in cardiac catheterization laboratories, primarily by cardiologists, interventional cardiologists, and cath lab professionals during coronary artery interventions.Q: What is the benefit of using a single-patient disposable PTCA kit?
A: Single-use design eliminates cross-contamination risks, streamlines setup, saves cleaning time, and ensures every component is consistently high in quality and sterility for accurate, safe, and efficient procedures.Q: How is patient safety ensured with the PTCA kit?
A: Each kit is CE certified, conforms to ISO 13485 standards, is latex-free to avoid allergic reactions, and comes sterilized with ETO, ensuring a high level of safety for every patient.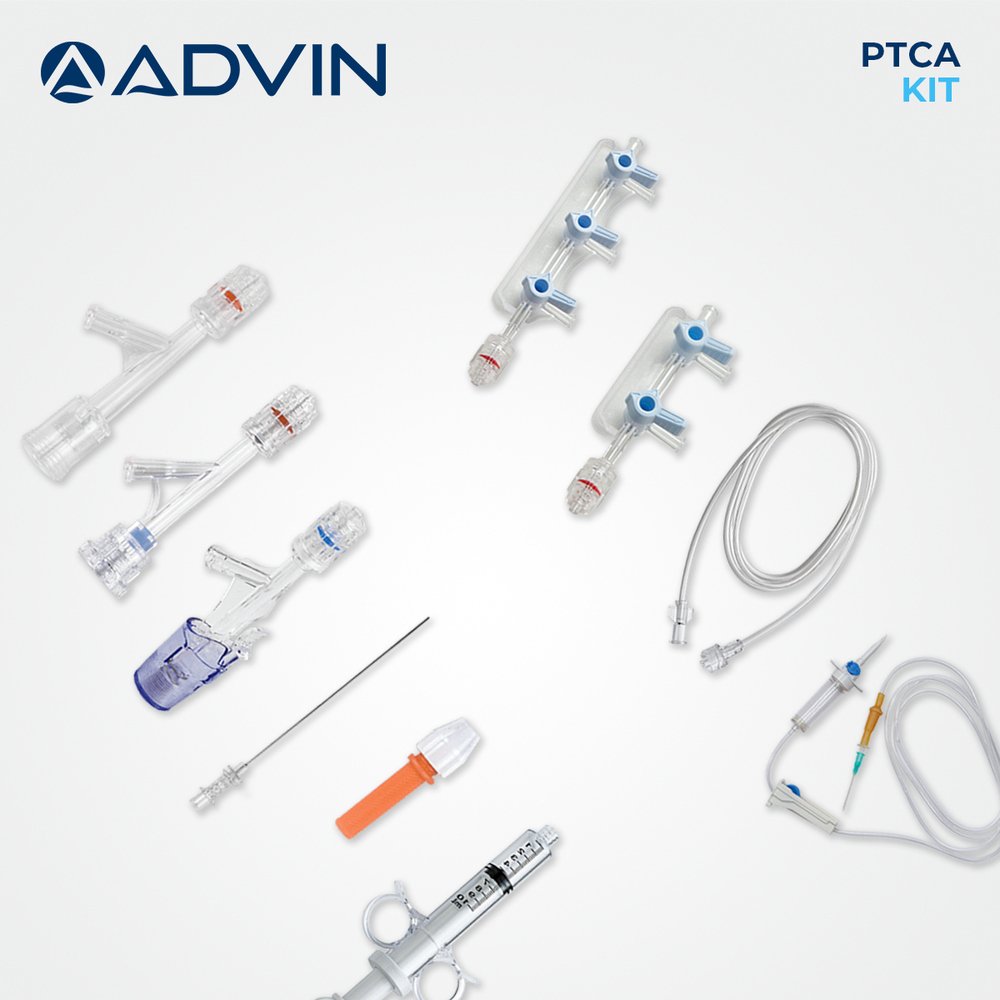




Price:
- 50
- 100
- 200
- 250
- 500
- 1000+

 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry



 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese