ICU Pressure Transducer
Price 9.7 USD ($)/ Piece
ICU Pressure Transducer Specification
- Accuracy
- 1 mmHg
- Storage Instructions
- Store in a cool, dry place below 40C
- Measurement Range
- -50 to +300 mmHg
- Features
- Sterile, High Accuracy, Fast Response, Secure Fluid Path, Air-elimination Vent
- Instruments Type
- Critical Care Monitoring
- Display Type
- No On-board Display (Connects to Patient Monitor)
- Shelf Life
- Up to 5 years (unopened and properly stored)
- Usage Type
- Medical/ICU Equipment
- Function
- Converts Physiological Pressure into Electrical Signal
- Equipment Type
- ICU Pressure Transducer
- Material
- Medical-Grade Polycarbonate, ABS, Silicone
- Condition
- New
- Technology
- MEMS (Microelectromechanical Systems) Sensor
- Portable
- Yes
- Wall Mounted
- No
- Real-Time Operation
- Yes
- Noise Level
- Low
- Operating Type
- Single Use, Disposable
- Use
- Invasive Blood Pressure Monitoring in ICUs
- Power Source
- External Monitor Interface (No Internal Power)
- Power Consumption
- Negligible
- Dimension (L*W*H)
- Approx. 8cm x 4cm x 2.5cm
- Weight
- Approx. 60g
- Color
- Transparent/White with Color-Coded Port
- Calibration
- Pre-calibrated, Zero Balance at Factory
- Packaging
- Individually Blister Packed
- Disposable
- Yes
- Connector Type
- Standard Luer Lock
- Sterility
- Ethylene Oxide Sterilized
- Tubing Length
- Approx. 100 cm
- Compliance
- CE, ISO 13485 Certified
- Temperature Range
- -20C to +55C (Storage)
- Latex Content
- Latex-free
- Transducer Interface
- Compatible with Most Major Patient Monitors
- Fluid Path Material
- DEHP-Free
About ICU Pressure Transducer
Advin Health Care is a trusted manufacturer of ICU Pressure Transducers designed to meet international quality standards. These devices are specifically engineered for continuous and accurate invasive pressure monitoring in intensive care units, ensuring reliable real-time data for critical patient management.
Key Features
- High-precision monitoring for critical care applications
- Real-time pressure signal conversion for accurate readings
- Stable and consistent performance in ICU environments
- Compatible with standard multi-parameter monitors
- Easy setup and user-friendly design
- Sterile, single-use option to enhance patient safety
Specifications
- Type: ICU Pressure Transducer
- Function: Invasive pressure monitoring (IBP)
- Material: Medical-grade components
- Sterilization: EO Sterilized / Non-sterile options available
- Usage: Single-use / Disposable
- Application: Arterial, venous, and central venous pressure monitoring in ICU
Certifications
CE, ISO 13485, and ISO 9001 certified
Product Keywords
ICU Pressure Transducer, IBP Transducer, Invasive Blood Pressure Sensor, Critical Care Monitoring Device, Medical Pressure Transducer
, , ,
Transductor de presin para UCI, transductor de presin arterial invasiva, sensor de presin arterial invasivo, dispositivo de monitorizacin de cuidados crticos, transductor de presin mdica
Capteur de pression pour soins intensifs, capteur de pression artrielle invasive, dispositif de surveillance des soins critiques, capteur de pression mdicale
High-Accuracy Monitoring for Critical Care
This transducer uses advanced MEMS sensor technology to ensure swift and precise conversion of physiological pressures into electrical signals. With secure DEHP-free fluid pathways and air-elimination venting, it offers dependable operation and safety for both patients and healthcare professionals. Its compatibility with most patient monitors and standardized luer lock connection streamlines ICU setup.
Safe, Convenient, and Hygienic Design
Each unit is factory pre-calibrated, sterilized with ethylene oxide, and individually blister-packed. The pressure transducer is latex-free and constructed from medical-grade polycarbonate, ABS, and silicone-ensuring minimal allergic reactions and maximum durability. Its disposable, single-use design reduces cross-contamination risks in intensive care environments.
Simplified Use and Maintenance
Designed for ease of use, this pressure transducer requires no additional calibration before use. Simply connect to the compatible patient monitor via the standard luer lock and color-coded port. The transparent and white housing allows easy visual inspection, and storage is hassle-free thanks to its compact size and extended shelf life.
FAQ's of ICU Pressure Transducer:
Q: How do I connect the ICU Pressure Transducer to a patient monitoring system?
A: The transducer features a standard Luer Lock connector and an interface compatible with most major patient monitors. Simply attach the device to the patient's line and connect the electrical port to the appropriate monitor input-no special adapters or calibration are required.Q: What are the storage requirements for the pressure transducer?
A: For optimal shelf life, store the unopened device in a cool, dry place below 40C. The sterile packaging and materials remain effective for up to five years under proper storage conditions.Q: When should I use this pressure transducer and what are its primary benefits?
A: This device is ideal for invasive blood pressure monitoring in critical care and ICU settings. Benefits include high accuracy (1 mmHg), fast response, reduced risk of contamination due to single-use design, and compatibility with a wide range of monitors.Q: Where is the product typically used within healthcare facilities?
A: The pressure transducer is primarily utilized in intensive care units (ICU) and other critical care environments where continuous and accurate blood pressure monitoring is needed for seriously ill patients.Q: What is the process for setting up the pressure transducer for use?
A: After verifying package integrity, remove the sterile transducer from its blister pack, connect it to the patient line using the Luer Lock, plug into the designated patient monitor port, and ensure the air-elimination vent is activated. The device is pre-calibrated and zero-balanced at the factory for immediate use.Q: Is the pressure transducer reusable, and how do I dispose of it?
A: No, this product is designed for single-use only to ensure maximum sterility and accuracy. After use, dispose of the transducer according to institutional protocols for medical waste.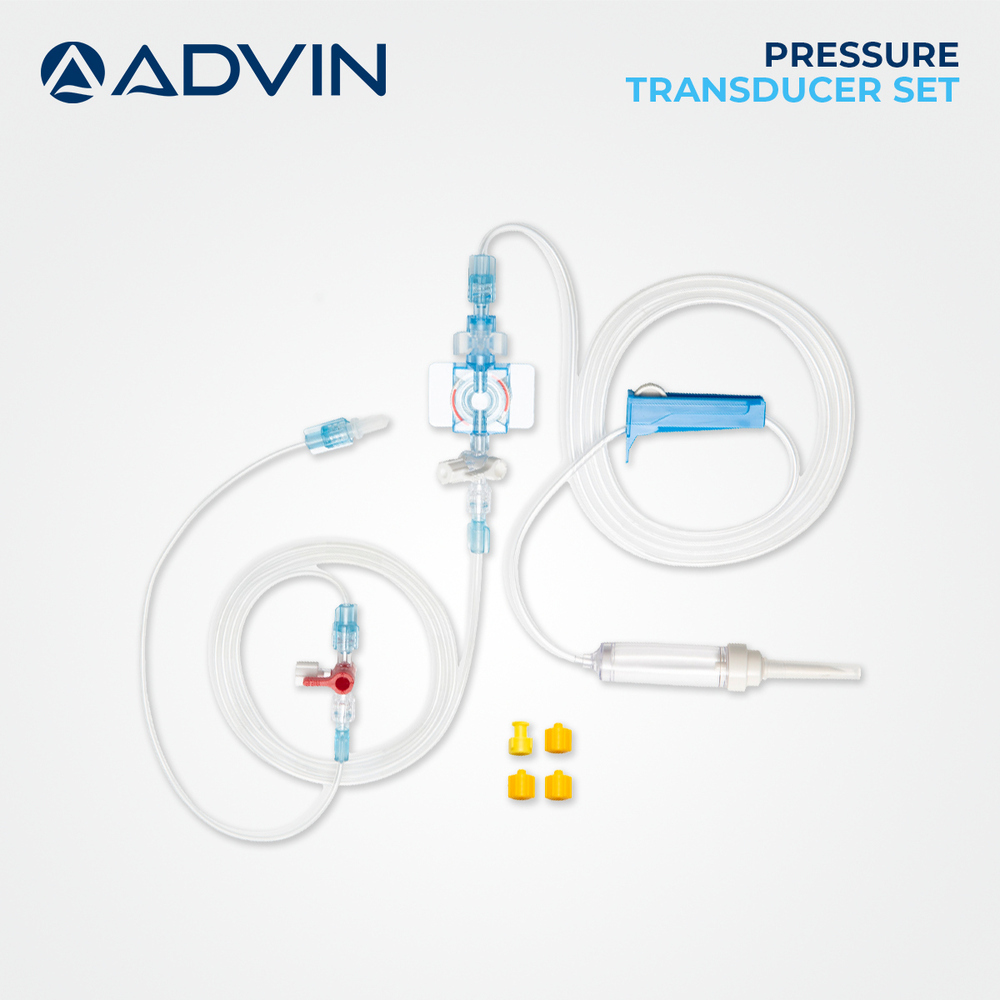

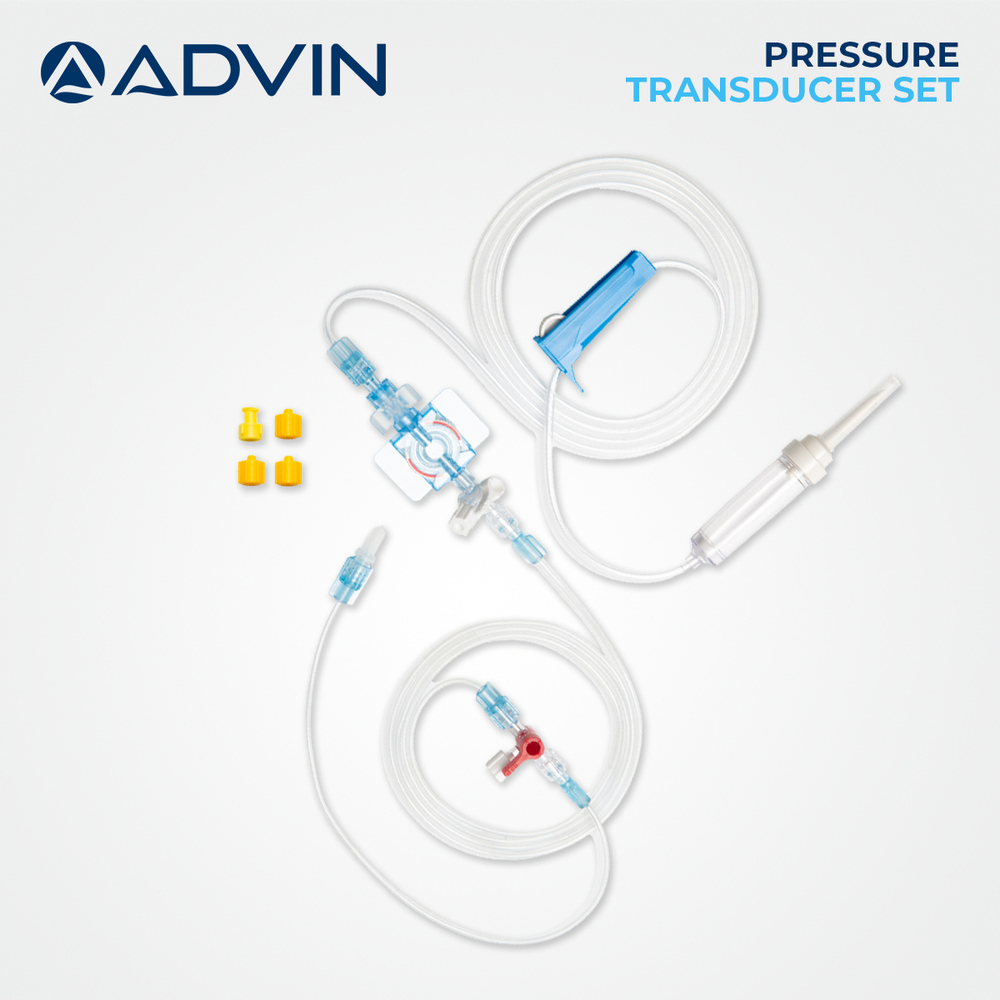


Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Cardiology Products Category
Cardiology Accessories - Medical Disposable Devices
Price 20 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Use : Cardiology Monitoring & Procedures
Operating Type : Manual
Condition : New
Real-Time Operation : Yes
Y Star PTCA Y Connector
Price 6.00 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Use : Cardiology, Angiography, PTCA Procedures
Operating Type : Manual
Condition : New
Real-Time Operation : Yes
Two Way Manifolds
Price 2 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Use : Hospital
Operating Type : Manual
Condition : New
Real-Time Operation : Yes

 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry




 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese