Percutaneous Guiding Catheter
Price 40 USD ($)/ Piece
Percutaneous Guiding Catheter Specification
- Function
- To provide access and guidance for intravascular interventions
- Storage Instructions
- Store in cool, dry place away from direct sunlight
- Measurement Range
- Available in various French sizes (5F, 6F, 7F etc.)
- Instruments Type
- Medical Catheter
- Accuracy
- High precision tip placement
- Features
- Atraumatic tip, radiopaque marker, flexible shaft, kink-resistant, latex-free
- Shelf Life
- 5 years
- Usage Type
- Single-use, Disposable
- Equipment Type
- Percutaneous Guiding Catheter
- Material
- Medical Grade Polyurethane / Polyethylene
- Condition
- New
- Technology
- Percutaneous Access
- Portable
- Yes
- Wall Mounted
- No
- Real-Time Operation
- Yes
- Noise Level
- Silent
- Operating Type
- Manual
- Use
- Cardiovascular Interventional Procedures
- Dimension (L*W*H)
- Varies by French Size, length typically 80-110 cm
- Weight
- Approx. 10-20 g per piece
- Color
- Blue / Transparent or Custom
- Compatibility
- Compatible with standard guidewires
- Packaging Type
- Sterile, individually packed
- Manufacturer Certification
- ISO 13485, CE Marked
- Catheter Lengths
- 80 cm, 90 cm, 100 cm, 110 cm
- French Size Options
- 5F, 6F, 7F, 8F
- Radiopacity
- Full length or tip-specific
- Tip Shape
- JL, JR, Amplatz, Multipurpose and others
- Intended Patient
- Adult and Pediatric
- Latex Content
- Latex-free
- Sterilization Type
- Ethylene Oxide (ETO) Sterilized
About Percutaneous Guiding Catheter
The percutaneous guiding catheter is designed to support precise control and stability during minimally invasive cardiovascular procedures. Its structure allows clinicians to maintain accurate positioning while advancing diagnostic or therapeutic devices. Advin Health Care manufactures this catheter with a focus on consistent quality, safety, and reliable clinical performance. The catheter supports efficient workflow and is suitable for a wide range of percutaneous cardiovascular interventions.
Advanced Features-
Designed for stable percutaneous vascular access
-
Supports smooth guidance of interventional devices
-
Compatible with standard guide wires and catheters
-
Intended for single-use clinical applications
Product Keywords
Percutaneous Guiding Catheter, Coronary Guiding Catheter, PTCA Guiding Catheter, Interventional Guiding Catheter, Angiography Guiding Catheter, Disposable Guiding Catheter
U3 aUUUU 1 UU U3 aUUUU UU UUU UaU U3 aUUUU UaU3U1 U UUU UaU 1U UU UU U3 aUUUU UUa Ua U1UU U3 aUUUU UaUU UU1U U UUU U3 aUUUU UU3a U UU U
NNo3412N1 12N2NNN 1 oNNN, o34N3412N12N1 12N2NNN 1 oNNN, 12N2NNN 1 oNNN N , 12NN212N 341212N1 12N2NNN 1 oNNN, 12N2NNN 1 oNNN N 123 343NN , 34 1234N342N1 12N2NNN 1 oNNN
CatAter guAa percutAneo, catAter guAa coronario, catAter guAa para PTCA, catAter guAa intervencionista, catAter guAa para angiografAa, catAter guAa desechable
CathAter guide percutanA, cathAter guide coronaire, cathAter guide pour angioplastie coronaire transluminale percutanAe (ACTP), cathAter guide interventionnel, cathAter guide pour angiographie, cathAter guide A usage unique
Versatile French Size and Tip Shape Options
This catheter is available in multiple French sizes (5F, 6F, 7F, 8F) and tip shapes (JL, JR, Amplatz, Multipurpose, and more), ensuring adaptability to various cardiovascular procedures and patient anatomies. Depending on procedural requirements, lengths ranging from 80 cm to 110 cm facilitate suitability for both adult and pediatric uses.
Safety-Focused, Sterile, and Certified
The catheter is individually packed and ethylene oxide (ETO) sterilized for safety. Its latex-free composition is ideal for sensitive patients, and manufacturer certifications-ISO 13485 and CE marking-underscore superior quality and global compliance. The product is strictly intended for single-use to minimize risks of infection.
Advanced Features for Effective Guidance
With an atraumatic tip, radiopaque marker for clear imaging, kink-resistant flexible shaft, and compatibility with standard guidewires, the catheter supports accurate real-time placement while minimizing trauma. These features increase procedural efficiency and patient safety during complex interventional cases.
FAQ's of Percutaneous Guiding Catheter:
Q: How is the Percutaneous Guiding Catheter used during cardiovascular interventions?
A: This catheter is inserted into the vascular system to provide a precise pathway for guidewires and other device delivery during cardiovascular interventional procedures. Its flexible, kink-resistant shaft and atraumatic tip allow for smooth navigation and accurate placement.Q: What French sizes and tip shapes are available for this catheter?
A: The catheter comes in French sizes 5F, 6F, 7F, and 8F, with a variety of tip shapes including JL (Judkins Left), JR (Judkins Right), Amplatz, Multipurpose, and others to suit specific anatomical and procedural requirements.Q: Where should the catheter be stored to maintain its quality?
A: Store the catheter in a cool, dry place away from direct sunlight. This helps preserve its shelf life of five years and ensures the sterile packaging remains intact until use.Q: What is the sterilization process for the catheter, and is it latex-free?
A: Each catheter is sterilized using ethylene oxide (ETO), ensuring it is free from microbial contamination. It is also latex-free, reducing the risk of allergic reactions in sensitive patients.Q: What are the benefits of the catheter's radiopaque features?
A: The radiopaque marker, present along the catheter's full length or at the tip, enhances visibility under fluoroscopy. This allows clinicians to precisely position the catheter during real-time interventions, improving procedural accuracy.Q: When is this catheter intended to be used, and is it reusable?
A: Designed for single-use only, the catheter should be used during one cardiovascular intervention and then safely disposed of to prevent cross-contamination and ensure optimal performance in each procedure.Q: What certifications and compliance standards does the catheter meet?
A: The catheter is manufactured under ISO 13485 standards and has CE marking, indicating compliance with international medical device safety and quality regulations.






Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Cardiology Products Category
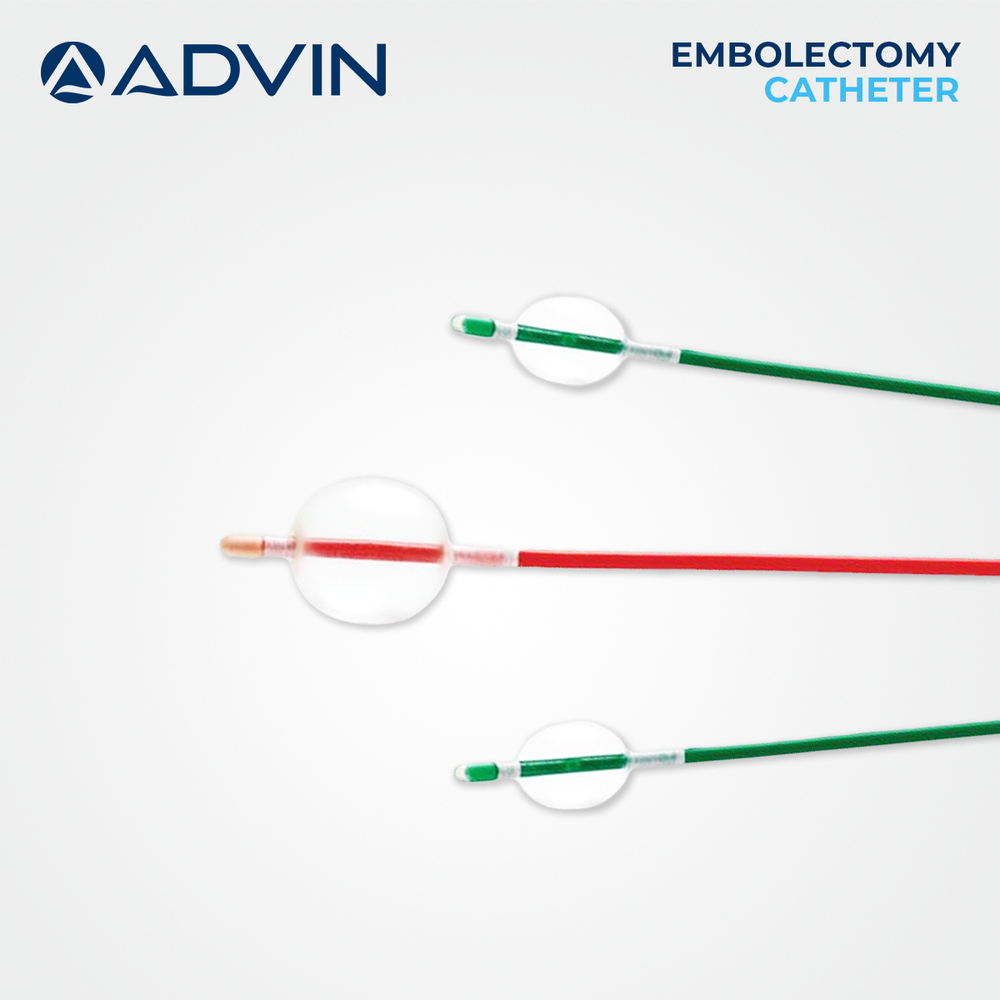
Endovascular Embolectomy Catheter
Price 30 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Features : Flexible shaft, radiopaque marker, hydrophilic coating, balloon tip design for clot retrieval
Shelf Life : 25 Years (as per manufacturer)
Material : Other, Medical Grade Polymer (such as Polyurethane or Nylon)
Usage Type : Single Use (Disposable)
Sterile Cardiology Products
Price 15 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Features : Sterile, Disposable, Precision Design
Shelf Life : 2 Years
Material : Other, Medical Grade Plastic and Stainless Steel
Usage Type : Single Use, Clinical
Cardiac Intervention Needle
Price 55 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Features : Sterile, sharp bevel, colorcoded hub, smooth surface, disposable
Shelf Life : 5 years
Material : Other, Medicalgrade Stainless Steel
Usage Type : Singleuse

 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry

 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese