PTCA Medical Kit
Price 15 USD ($)/ Piece
PTCA Medical Kit Specification
- Shelf Life
- 3-5 years (sealed and stored properly)
- Features
- Comprehensive kit with guidewire, introducer, syringes, dilators, Y-connector, torquer
- Storage Instructions
- Store in cool, dry place away from direct sunlight
- Accuracy
- High precision components for clinical use
- Measurement Range
- Catheters: 4-8 Fr, Guidewire: 0.014-0.038 inch
- Usage Type
- Single-use, disposable
- Instruments Type
- Medical Kit
- Function
- Facilitate minimally invasive balloon angioplasty
- Equipment Type
- PTCA Medical Kit
- Material
- Medical-grade polymer, stainless steel, silicone
- Condition
- New
- Technology
- Catheterization-based angioplasty procedure kit
- Portable
- Yes
- Wall Mounted
- No
- Real-Time Operation
- Yes
- Noise Level
- Silent Operation
- Operating Type
- Manual
- Use
- Cardiovascular procedure - Percutaneous Transluminal Coronary Angioplasty
- Dimension (L*W*H)
- Variable (customizable kit sizes)
- Weight
- Approx. 200-500 grams (kit dependent)
- Color
- Transparent/White/Blue (depends on component)
- Balloon Compatibility
- Yes (designed for angioplasty balloons)
- Latex Free
- Yes, latex free
- Kit Packaging
- Sealed, individually packed tray
- Guidewire Size
- 0.014", 0.018", 0.035", 0.038"
- Regulatory Approval
- CE certified, ISO 13485 compliant
- Intended Patient
- Adults (consult manufacturer for pediatric use)
- Sterility
- Sterile, EO gas sterilized
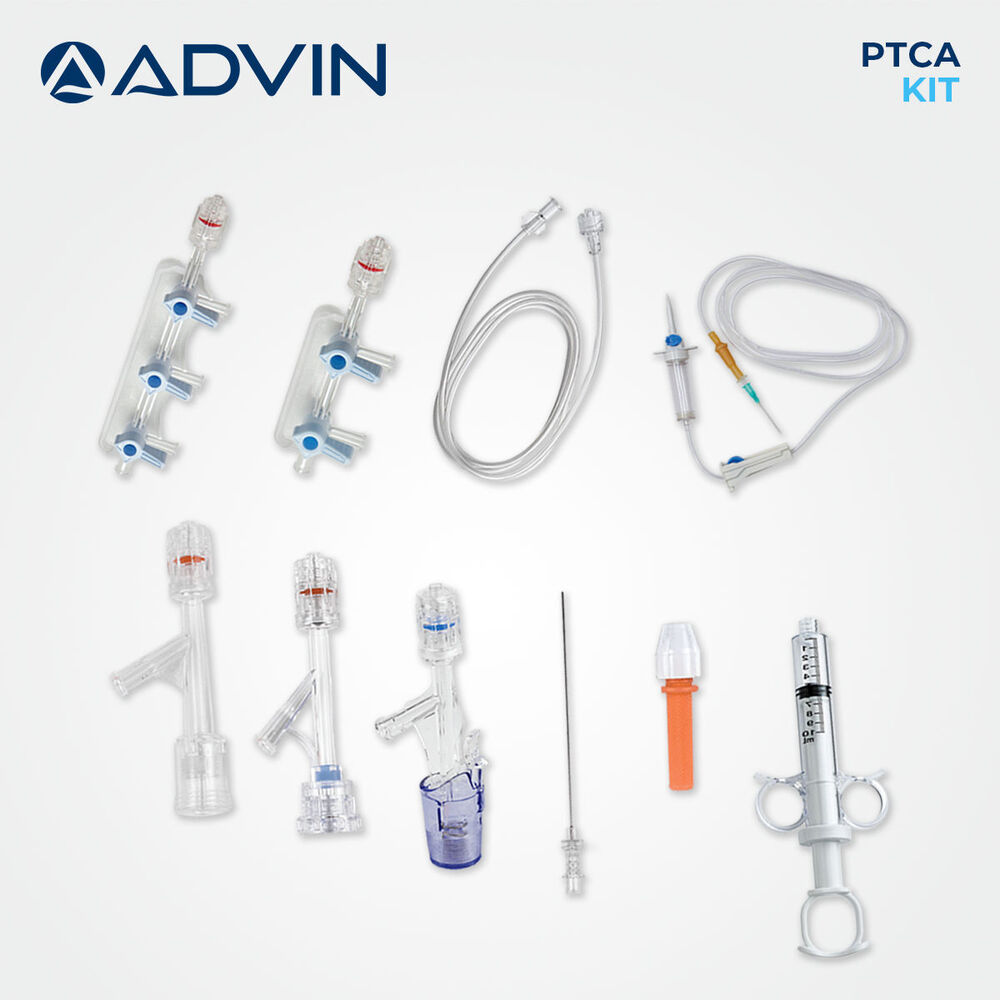
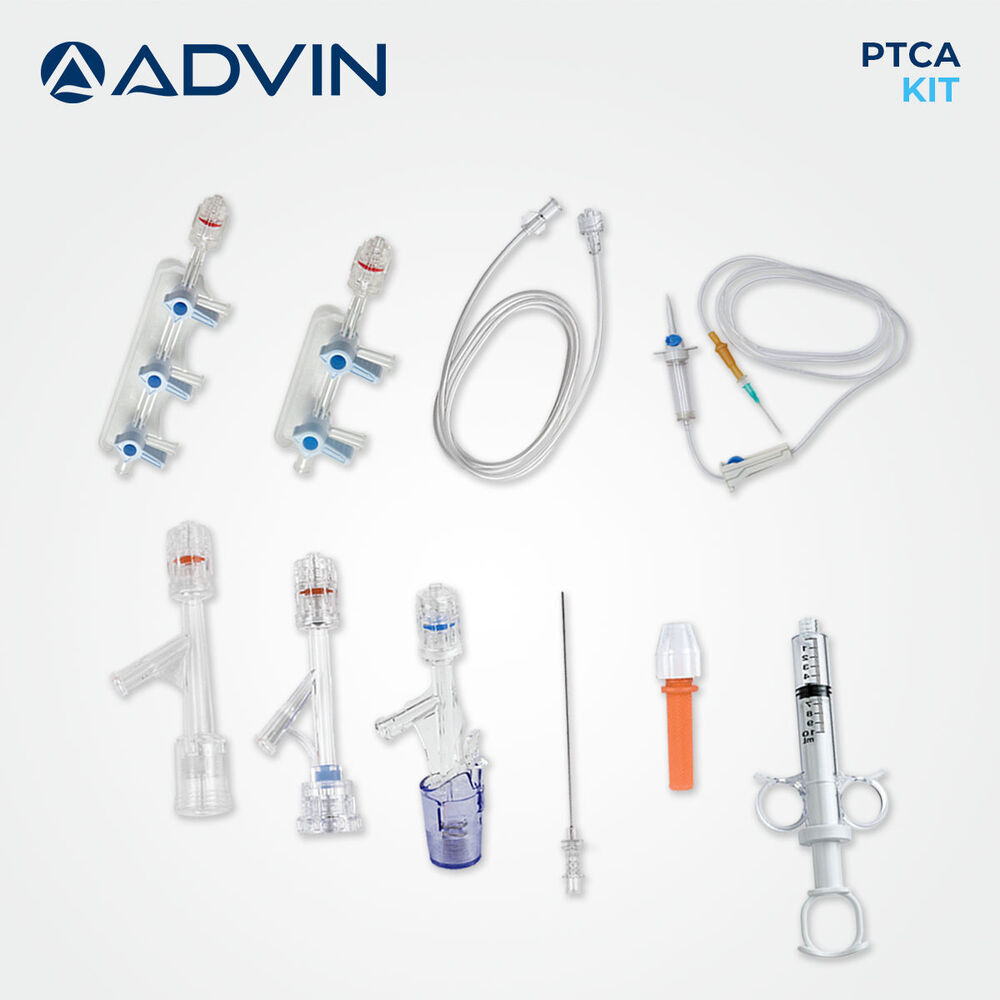
- Kit Components
- Guidewire, Introducer Sheath, Y-Connector, Tuohy-Borst Adapter, Dilator, Retrieval Device, Injection Syringe, Torquer, Hemostasis Valve
- Catheter Size Compatibility
- 4-8 Fr
About PTCA Medical Kit
Advin Health Care is a trusted manufacturer of PTCA Medical Kits designed to meet international quality standards. These kits are used in percutaneous coronary interventions for effective management of coronary artery disease.
Key Features
- Comprehensive kit for percutaneous transluminal coronary angioplasty (PTCA)
- Includes guide wire, balloon catheter, Y-connector, and accessory components
- High-quality, durable materials for reliable performance
- Designed for smooth device navigation and accurate deployment
- Compatible with standard angioplasty equipment and monitoring systems
Certifications
We are certified with CE, ISO 13485, and ISO 9001 standards.
Product Keywords
PTCA Medical Kit, Coronary Angioplasty Kit, Percutaneous Coronary Intervention Kit, PTCA Kit, Interventional Cardiology Kit, Angioplasty Procedure Kit
UUU1 Ua U UaU3U1 U UUU UaU 1U UU UU UUU1 Ua UU1U UaU UUU1 Ua Ua U UaU 1U UU UU UUU1 Ua UU1U UaU 1U UU UU UUU1 Ua UUU Ua UU UUU1 Ua UU1U
N 12No 1 1234N N , 1234N N o34N3412N12341 123 34NN o , 1234N N NNNo3412341 o34N3412N12341 12NN212N , 1234N N , 1234N N 12NN212N 341212341 oN 34343 , 1234N N N342 12 N 123 34NN o .
Kit mAdico para PTCA, kit de angioplastia coronaria, kit de intervenciA3n coronaria percutAnea, kit de PTCA, kit de cardiologAa intervencionista, kit de procedimiento de angioplastia
Kit mAdical PTCA, kit d'angioplastie coronaire, kit d'intervention coronaire percutanAe, kit PTCA, kit de cardiologie interventionnelle, kit pour procAdure d'angioplastie
Comprehensive Angioplasty Solution
The PTCA Medical Kit assembles all necessary tools for percutaneous transluminal coronary angioplasty in one complete, single-use package. Each component, from the high-precision guidewires to secure introducer sheaths and torquer, is crafted to streamline the angioplasty process and ensure optimal safety. The kit features EO gas sterilization and adheres to CE and ISO 13485 standards, providing confidence in sterility and quality for each procedure.
Versatile Compatibility and Precision
With compatibility for catheter sizes 4-8 Fr and guidewires ranging from 0.014" to 0.038", this kit supports a wide array of clinical scenarios. The carefully selected contents enable tailored, patient-specific procedures, helping clinicians achieve high-precision outcomes in adult angioplasty cases. The balloon compatibility and inclusion of crucial components further ease the process and enhance usability.
Safe, Sterile, and Convenient Design
Every kit is latex-free and sterilized using EO gas, minimizing risks and ensuring patient safety. The sealed, portable packaging preserves kit sterility with a three-to-five year shelf life when stored properly. All materials used are medical-grade, and the transparent or color-coded elements facilitate easy identification and usage during critical cardiovascular procedures.
FAQ's of PTCA Medical Kit:
Q: How should the PTCA Medical Kit be used during angioplasty?
A: The PTCA Medical Kit is designed for use during percutaneous transluminal coronary angioplasty (PTCA). All instruments, including the guidewire, introducer sheath, dilator, balloon-compatible Y-connector, and hemostasis valve, are used to perform minimally invasive balloon angioplasty on adult patients. A skilled clinician should follow standard angioplasty protocols for safe and effective kit usage.Q: What catheter and guidewire sizes are compatible with this kit?
A: This kit supports catheter sizes ranging from 4 to 8 French (Fr) and guidewires of 0.014", 0.018", 0.035", and 0.038" diameters, making it versatile for various angioplasty needs. The components are specifically chosen to ensure compatibility and ease of integration for a range of clinical scenarios.Q: When should this kit be selected for use?
A: The PTCA Medical Kit is ideal for use in adult cardiovascular procedures requiring percutaneous transluminal coronary angioplasty. It is most suitable when a comprehensive, sterile, and single-use solution is needed, especially in clinics and hospitals prioritizing efficient workflow and high safety standards.Q: Where should the PTCA Medical Kit be stored before use?
A: It should be stored in a cool, dry location, away from direct sunlight, to maintain its sterility. The kit remains effective for three to five years when properly stored in its sealed, individually packed tray.Q: What is the benefit of using a single-use, EO-sterilized angioplasty kit?
A: Using a single-use, EO gas-sterilized kit reduces the risk of cross-contamination and infection, supports consistent clinical outcomes, and ensures that every component is ready and safe for immediate use in sterile environments.Q: How does the kit support real-time and precise cardiovascular procedures?
A: All components are engineered to deliver high precision and reliability during real-time cardiovascular interventions. Transparent or color-coded, each tool is designed for smooth integration into existing protocols, supporting clinicians in delivering accurate and efficient care.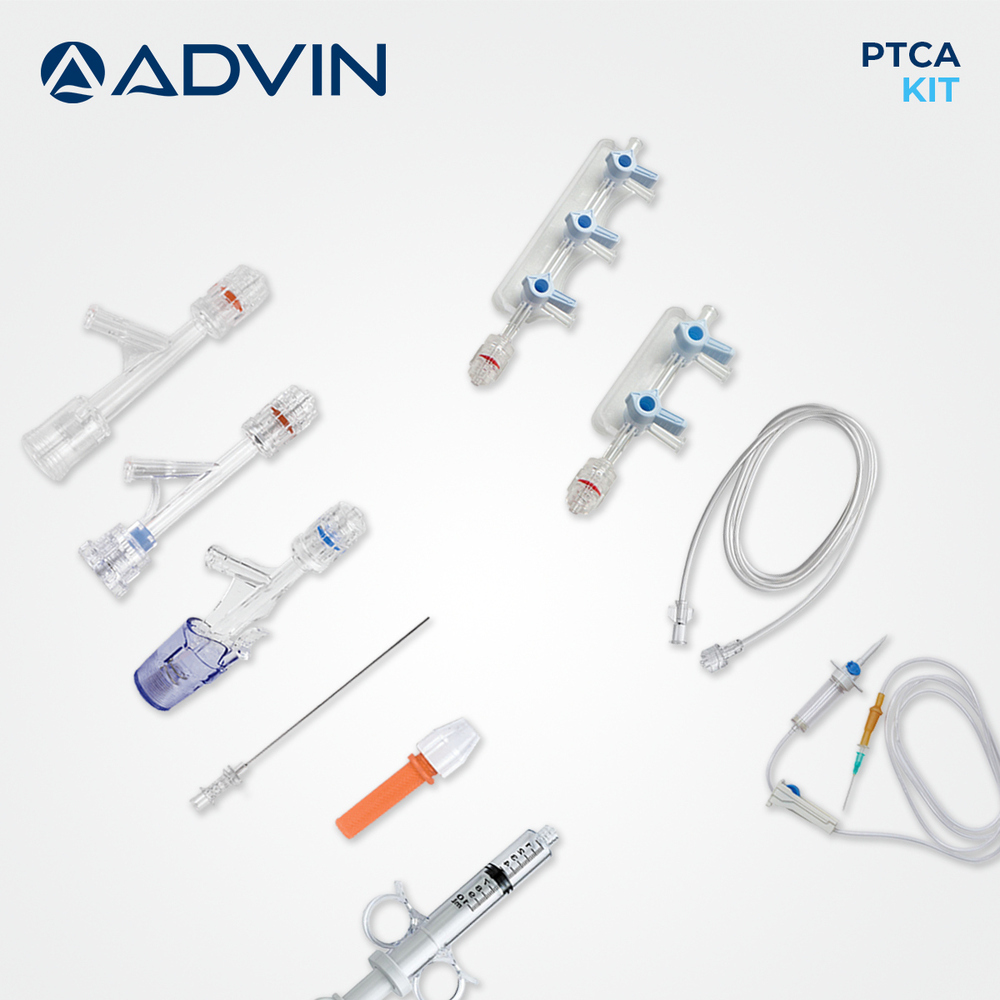




Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Cardiology Products Category
Coronary angioplasty and stents (PCI)
Price 120 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Wall Mounted : No
Condition : New
Operating Type : Other, Manual/Assisted
Real-Time Operation : Yes
Pressure Monitoring Line
Price 0.25 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Wall Mounted : No
Condition : New
Operating Type : Manual
Real-Time Operation : Yes

 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry




 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese