Surgical Area Cover
Price 5.10 USD ($)/ Piece
Surgical Area Cover Specification
- Shelf Life
- 5 Years
- Usage Type
- Single Use
- Storage Instructions
- Store in a cool, dry place, away from direct sunlight
- Instruments Type
- Medical Disposable
- Function
- Provides an aseptic barrier in surgical environments
- Features
- Sterile, Disposable, Fluid Resistance, Tear Resistant, Soft Texture, Lint Free
- Equipment Type
- Surgical Area Cover
- Material
- SMS (Spunbond-Meltblown-Spunbond) Non Woven Fabric
- Condition
- New
- Technology
- Non-woven Fabrication
- Portable
- Yes
- Wall Mounted
- No
- Real-Time Operation
- No
- Operating Type
- Manual
- Use
- Used to cover and isolate the surgical area during medical procedures
- Dimension (L*W*H)
- As per requirement / Customizable
- Weight
- Lightweight
- Color
- Blue
- Edge Type
- Ultrasonically Sealed Edges
- Tear Resistance
- High
- Packaging Type
- Sterile Packed
- Pattern
- Plain or with Adhesive Fenestration
- CE Certified
- Yes
- Water Resistance
- Yes
- Sterilization Method
- Ethylene Oxide (ETO) or Gamma Radiation
About Surgical Area Cover
Nivda Drapes is globally leading manufacturer and supplier of Surgical Area Cover. Surgical Area Cover is a sterile disposable drape used to protect the surgical site and surrounding surfaces during medical procedures. It helps maintain a clean and controlled environment in hospitals and operating rooms. These covers support infection control and improve hygiene during surgical procedures.Advanced Features:
Sterile disposable cover designed for surgical area protection
Helps maintain a clean and controlled operative field
Fluid-resistant material for effective barrier protection
Easy placement around the surgical site
Single-use design to support hygiene and infection control
Surgical Area Cover, Sterile Surgical Cover, Disposable Surgical Cover, Medical Area Cover, OT Area Cover, Non Woven Surgical Cover, Hospital Surgical Drapes
غطاء منطقة العمليات الجراØية، غطاء جراØÙŠ معقم، غطاء جراØÙŠ للاستخدام مرة واØدة، غطاء منطقة طبية، غطاء منطقة غرÙØ© العمليات، غطاء جراØÙŠ غير منسوج، أغطية جراØية للمستشÙيات
ХирургичеÑкое покрывало, Ñтерильное хирургичеÑкое покрывало, одноразовое хирургичеÑкое покрывало, медицинÑкое покрывало, покрывало Ð´Ð»Ñ Ð¾Ð¿ÐµÑ€Ð°Ñ†Ð¸Ð¾Ð½Ð½Ð¾Ð¹, нетканое хирургичеÑкое покрывало, больничные хирургичеÑкие проÑтыни
Cubierta para área quirúrgica, cubierta quirúrgica estéril, cubierta quirúrgica desechable, cubierta para área médica, cubierta para área de quirófano, cubierta quirúrgica no tejida, campos quirúrgicos hospitalarios
Champ opératoire stérile, champ opératoire stérile, champ opératoire jetable, champ médical, champ opératoire non tissé, draps chirurgicaux hospitaliers
Optimal Protection in Surgical Settings
This surgical area cover is meticulously crafted to create a reliable aseptic barrier during medical procedures. Its water-resistant, fluid-proof SMS non-woven fabric ensures the surgical area remains uncontaminated, safeguarding both patients and healthcare professionals. The product's sterilization-via ETO or Gamma Radiation-further prevents microbial transmission.
Designed for Efficiency and Comfort
With options for plain or adhesive fenestration patterns and ultrasonically sealed edges, the cover adapts to various surgical needs. Its lightweight, lint-free material is soft on the skin yet highly tear-resistant, ensuring comfort and minimal disruption during use. Customizable dimensions provide flexibility for diverse operating environments.
Trusted Certification and Quality
CE certification and rigorous manufacturing standards confirm the product's safety and reliability. Packaged in a sterile manner, it ensures immediate usability and extended shelf life. As a disposable item, it eliminates cross-contamination risks and aids efficient post-surgery cleanup for healthcare teams.
FAQ's of Surgical Area Cover:
Q: How is the surgical area cover sterilized before use?
A: The surgical area cover is sterilized using either Ethylene Oxide (ETO) or Gamma Radiation, ensuring it remains free from bacteria and safe for use in aseptic medical environments.Q: What are the main benefits of using an SMS non-woven fabric cover during surgery?
A: SMS (Spunbond-Meltblown-Spunbond) non-woven fabric offers excellent fluid resistance and tear resistance, is lint-free, lightweight, and soft, providing both patient protection and comfort during procedures.Q: When should the surgical area cover be used during medical procedures?
A: It should be applied before starting any sterile medical or surgical procedure to establish a protective barrier around the operative site and maintain sterility throughout the process.Q: Where can the surgical area cover be customized for size or fenestration?
A: These covers are available with customizable dimensions and offer options for either a plain or adhesive fenestration pattern, suitable for various surgical settings and requirements.Q: What is the recommended process for storing the surgical area covers?
A: Store the covers in a cool, dry environment away from direct sunlight to preserve their sterility and effectiveness, ensuring optimal performance up to their five-year shelf life.Q: How does ultrasonically sealed edge technology improve the safety of the cover?
A: Ultrasonically sealed edges prevent fiber dispersal and potential contamination, ensuring a secure, intact barrier throughout the procedure.Q: What are the usage instructions and disposal recommendations for this product?
A: The cover is for single-use only. After the procedure, dispose of it according to local guidelines for medical waste to prevent cross-contamination and maintain a sterile environment.






Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Disposable Drapes Category
Infection Prevention in Surgery
Price 9.60 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Dimension (L*W*H) : Standard kit size, 35 cm x 22 cm x 8 cm (approx.)
Weight : 1.2 kg (approx.)
Color : Blue/White (as per standard medical supplies)
Portable : Yes
IVF Drape Kit
Price 9.02 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Dimension (L*W*H) : Variable (Kit includes multiple sizesStandard approx. 100 x 120 cm drapes)
Weight : Approximately 300500 g (Complete Kit)
Color : Blue
Portable : Yes
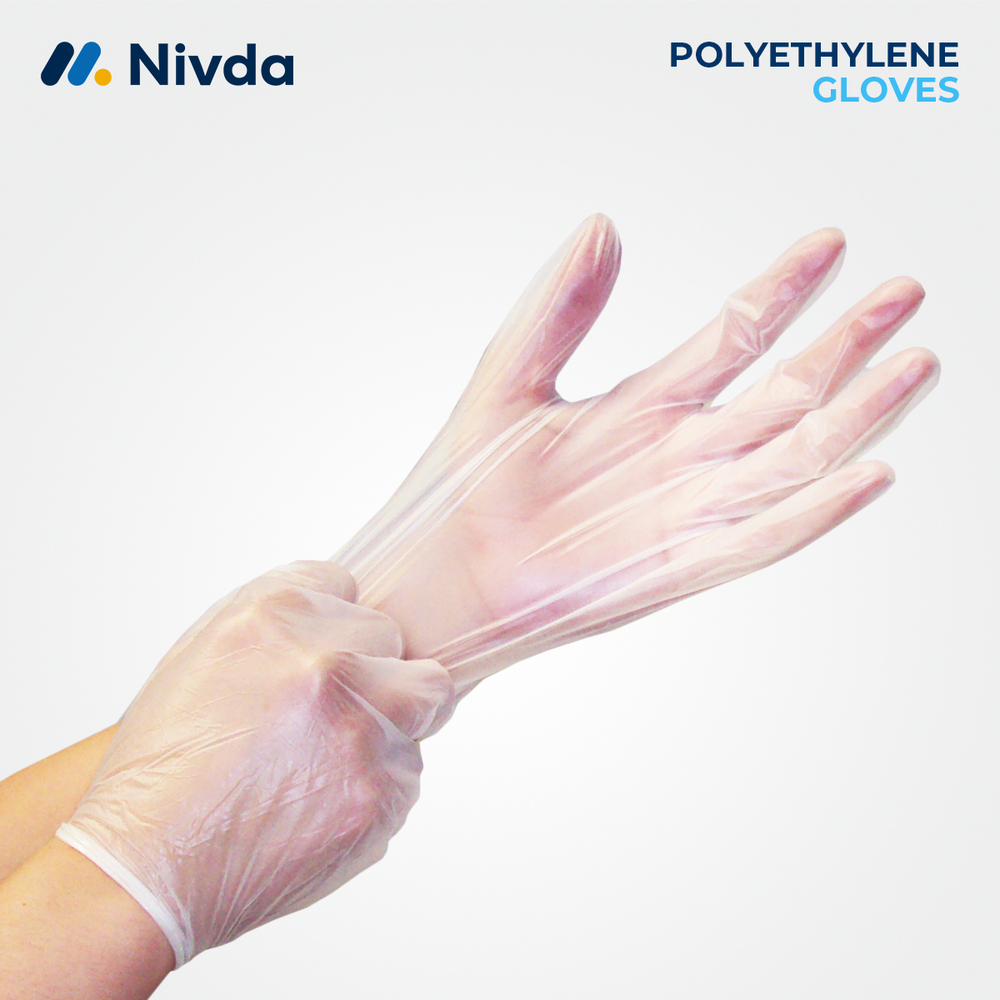
Polyethylene Gloves 35 GSM Ex. Large
Price 3.20 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Dimension (L*W*H) : Ex. Large (Fits Most Adult Hands)
Weight : Lightweight
Color : Transparent
Portable : Yes
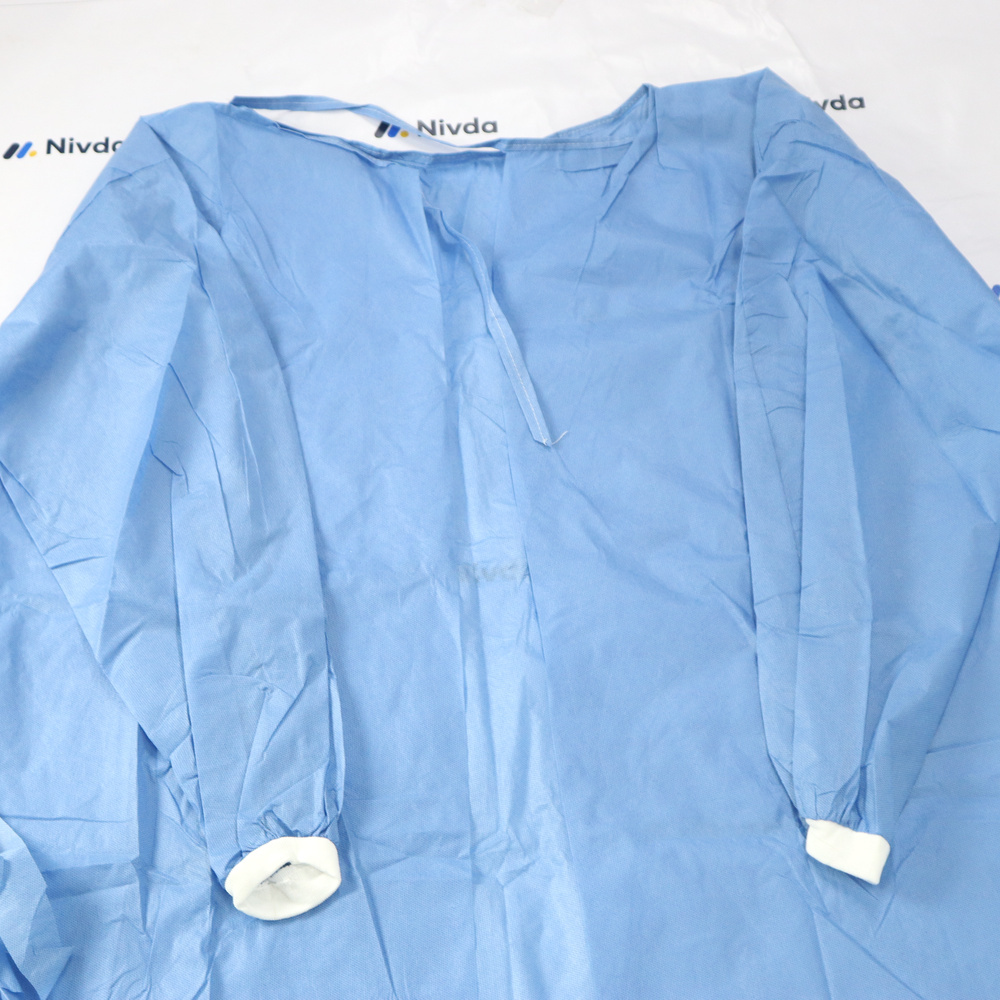
SMS 45 GSM Surgeon Gown
Price 2.76 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Dimension (L*W*H) : Standard Adult Size
Weight : Lightweight
Color : Blue
Portable : Yes

 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry


 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese