Hip Implants
Price 3 USD ($)/ Piece
Hip Implants Specification
- Shelf Life
- 5-10 years (Sterile, Unused)
- Instruments Type
- Implant
- Function
- Restore hip joint mobility
- Storage Instructions
- Store in a sterile, dry environment
- Usage Type
- Medical/Hospital
- Features
- Corrosion resistance, High durability, Biocompatible, Smooth surface finish
- Accuracy
- High fit precision
- Measurement Range
- Multiple sizes available
- Equipment Type
- Hip Implants
- Material
- Stainless Steel / Titanium Alloy
- Condition
- New
- Technology
- Orthopedic Precision Engineering
- Portable
- Yes
- Wall Mounted
- No
- Operating Type
- Surgical
- Use
- Hip Joint Replacement Surgery
- Power Source
- Manual
- Dimension (L*W*H)
- Standard/Custom Sizes
- Weight
- Variable per design
- Color
- Metallic/Silver
- Radiolucent Markers
- Present
- Application Area
- Total Hip Arthroplasty (THA)/Partial Hip Replacement
- Component Type
- Femoral Stem, Acetabular Cup, Liner, Head
- Surface Coating
- Polished/Porous (optional)
- Articulation Type
- Ball-and-socket
- Packaging Type
- Blister/Sealed Pack
- Sterility
- Sterile pack supplied
- ISO Certification
- ISO 13485 approved
- Compatibility
- Compatible with major hip system instruments
- Manufacturers Warranty
- Available upon request
- Sterilization
- EO Gas/Autoclave compatible
About Hip Implants
Hip Implants are widely used in orthopedic trauma surgeries for conditions such as femoral neck fractures, arthritis, and joint degeneration. They provide stable support and facilitate smooth joint movement, helping patients regain functional mobility quickly. Advin Health Care manufactures Hip Implants using medical-grade materials, ensuring strength, biocompatibility, and long-term durability. Available in various designs and sizes, these implants cater to different surgical needs, offering optimal clinical outcomes and improved patient quality of life.
Advanced Features:
-
Made from premium medical-grade materials for strength and biocompatibility
-
Available in multiple designs and sizes to suit patient anatomy
-
Restores mobility and reduces pain in damaged hip joints
-
Provides stable and durable support for long-term use
-
Suitable for trauma, arthritis, and reconstructive hip surgeries
Advanced Materials and Engineering
Utilizing high-quality stainless steel or titanium alloy, our hip implants ensure exceptional strength, biocompatibility, and long-term durability. The implants are forged using precise orthopedic engineering, accommodating a range of patient anatomies through standard and custom dimensions.
Optimal Surgical Compatibility
Designed for seamless integration, the implants fit with most leading hip system instruments. This compatibility assures surgeons of smooth operative procedures in both total and partial hip replacement surgeries, improving workflow and procedural efficiency.
Safety and Sterility Guaranteed
Every hip implant comes in a sterile, blister or sealed pack, having undergone rigorous EO Gas or autoclave sterilization. Compliance with ISO 13485 standards assures consistently high safety, sterility, and quality from manufacturing to surgical application.
FAQ's of Hip Implants:
Q: How are these hip implants sterilized before supply?
A: The hip implants are sterilized utilizing either Ethylene Oxide (EO) Gas or autoclave processes. After sterilization, each implant is supplied in a securely sealed sterile blister pack to maintain its sterility until use.Q: What components are included in your hip implant system?
A: Our system includes key hip implant components: femoral stem, acetabular cup, liner, and head, all designed for ball-and-socket articulation and compatible with major hip system instruments used in hip replacement surgeries.Q: When should these hip implants be used?
A: These implants are intended for use during total hip arthroplasty (THA) or partial hip replacement procedures, particularly for patients with damaged or diseased hip joints requiring surgical restoration of mobility.Q: Where can these hip implants be stored before surgery?
A: The implants should be stored in a sterile and dry environment as specified in the storage instructions. This guarantees maintained sterility and extends the shelf life to up to 5-10 years, provided the packaging remains unopened.Q: What benefits do radiolucent markers provide in these implants?
A: Radiolucent markers allow clinicians to accurately visualize implant positioning during and after surgery using X-ray imaging, ensuring correct alignment and enhancing postoperative assessment.Q: How does the surface coating option affect implant performance?
A: Implants come with either a polished surface for smooth articulation or an optional porous coating to encourage bone in-growth and fixation, allowing customization based on surgical requirements and patient needs.Q: What warranty and certification support do you offer?
A: A manufacturer's warranty is available upon request, and all implants are approved under ISO 13485 certification, ensuring stringent quality control and compliance with international medical device standards.



Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Orthopedic Products Category
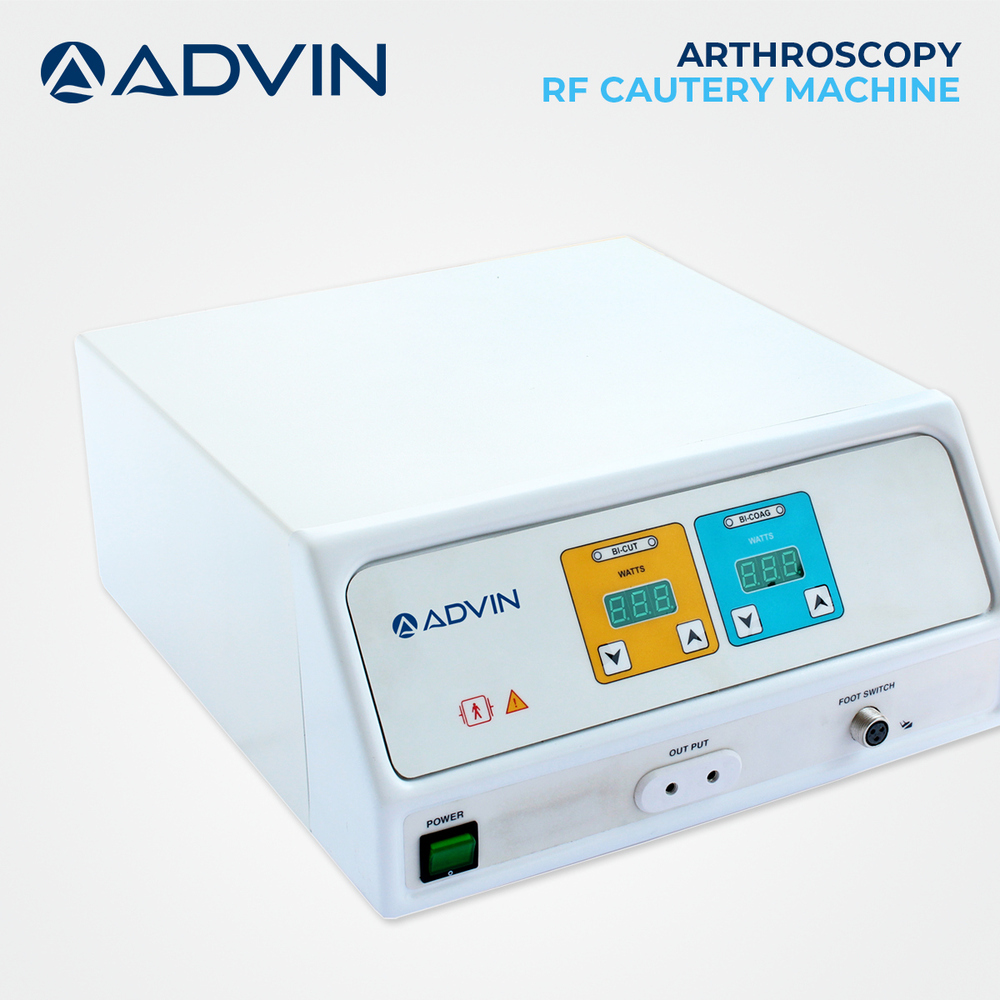
Arthroscopy RF Cautery Machine
Price 812 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Portable : Yes
Storage Instructions : Store in clean, dry place away from moisture
Color : White & Blue
Equipment Type : Arthroscopy RF Cautery Machine
Spinal Fixation System
Price 1 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Portable : Yes
Storage Instructions : Store in cool, dry place away from direct sunlight
Color : Metallic / Silver
Equipment Type : Spinal Fixation System
Interbody Fusion Cage
Price 6 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Portable : Yes
Storage Instructions : Keep in dry, sterile conditions at recommended temperature
Color : Metallic (Silver/Grey)
Equipment Type : Interbody Fusion Cage
Tibia Interlocking Nail
Price 3 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Portable : Yes
Storage Instructions : Store in a Dry, Sterile Environment
Color : Metallic Silver
Equipment Type : Orthopedic Surgical Implant

 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry



 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese