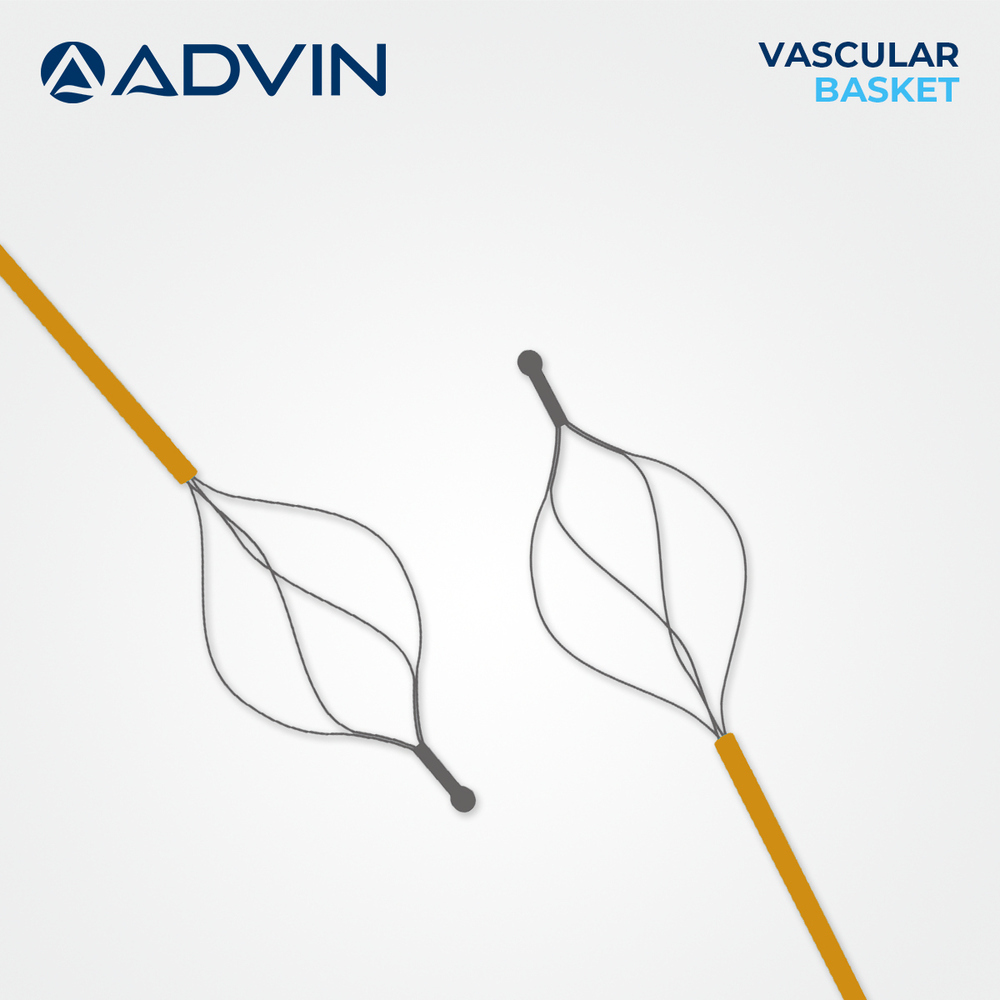
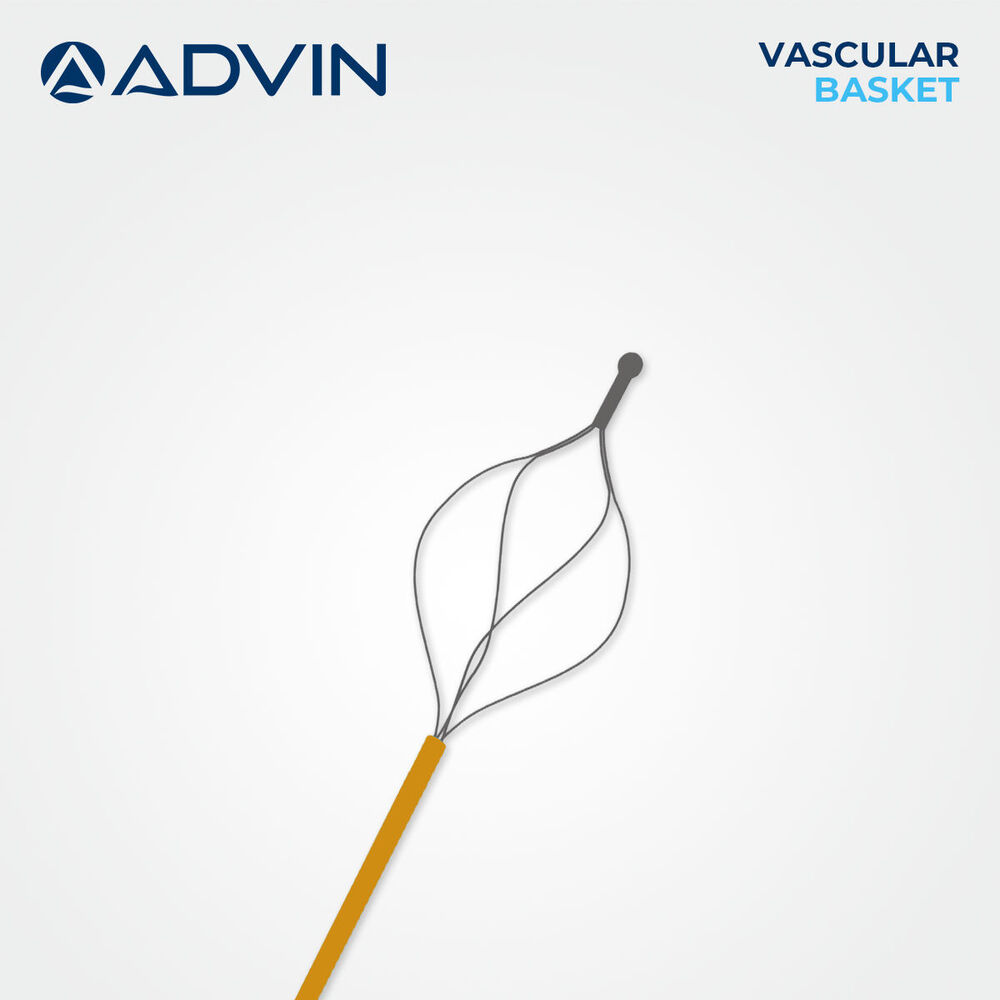
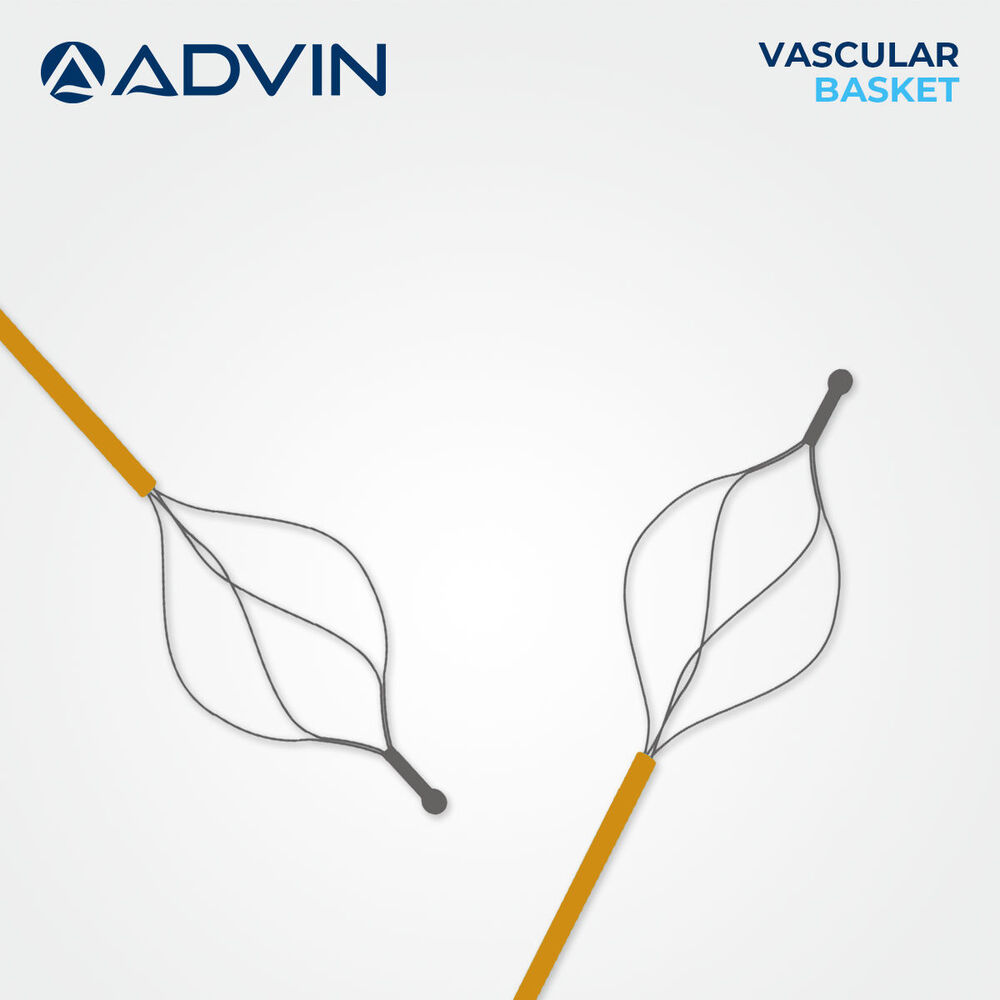
Vascular Basket Retrieval Device
Price 60 USD ($)/ Piece
Vascular Basket Retrieval Device Specification
- Function
- Retrieves Foreign Objects from Blood Vessels
- Measurement Range
- Basket Diameter 2-4 mm
- Features
- Radiopaque, Flexible, Kink Resistant, Precise Control
- Accuracy
- High Retrieval Success Rate
- Shelf Life
- 5 Years
- Instruments Type
- Endovascular Retrieval Device
- Usage Type
- Single Use (Disposable)
- Storage Instructions
- Store in Cool, Dry Place
- Equipment Type
- Vascular Basket Retrieval Device
- Material
- Medical Grade Stainless Steel and Nitinol
- Condition
- New
- Technology
- Minimally Invasive Endovascular Technology
- Portable
- Yes
- Wall Mounted
- No
- Real-Time Operation
- Yes
- Noise Level
- Silent Operation
- Operating Type
- Manual
- Use
- Vascular Foreign Body Retrieval
- Dimension (L*W*H)
- Varies by Model (e.g. 1200 mm length, 2-4 mm basket diameter)
- Weight
- Lightweight
- Color
- Silver/Metallic
- Regulatory Compliance
- CE Marked, ISO 13485 Certified
- Radiopacity
- Radiopaque Tip and Basket for Fluoroscopic Guidance
- Sterility
- Sterile, EO (Ethylene Oxide) Sterilized
- Flexibility
- High Flexibility for Navigation in Tortuous Anatomy
- Packaging
- Individually Packed in Sterile Blister Pack
- Handle Type
- Ergonomic, Non-Slip
- Clinical Application
- Removal of Thrombus, Calculi, or Other Intravascular Debris
- Basket Type
- 4-Wire or 6-Wire Construction
- Compatibility
- Suitable for Guide Catheter Sizes 4 Fr-7 Fr
About Vascular Basket Retrieval Device
The vascular basket retrieval device is designed to support precise and reliable removal of embolic material during complex vascular interventions. Its basket mechanism enables secure engagement while helping minimize trauma to the vessel wall. Advin Health Care manufactures this device with a strong focus on clinical safety, consistency, and dependable performance. The device is widely used in interventional radiology and vascular procedures, supporting efficient workflow and confident embolism management.
Advanced Features- Basket design for controlled embolus and foreign body retrieval
-
Supports safe navigation within vascular pathways
-
Designed for use in endovascular procedures
-
Intended for single-use clinical applications
Certifications
We are certified with CE, ISO 13485, and ISO 9001 standards.
Product Keywords
Vascular Basket Retrieval Device, Nitinol Vascular Basket, Embolus Retrieval Device, Thrombus Capture Device, Interventional Vascular Basket, Coronary and Peripheral Retrieval Device
U2 3a UU1U U UUU 3U UU1U U UUU UUUU1 UU UUUaUUUU U2 3a UUa U2 UaU Ua 3U UU1U U UUU Ua UU U2 3a U UUU UaU UUUUU
NNN341NN234 N 2N12 N N34NN 342 2 2 12 N 1234342341 o34N 12o , NNNN341NN234 N 2N12 N N1434342, NNNN341NN234 N N2N NN3414342, 12NN212N 341212N N34NN NNN o34N 12o, NNNN341NN234 N 2N12 N o34N3412N12NN N NN NNo N NNN 1.
Dispositivo de recuperaciA3n de cesta vascular, cesta vascular de nitinol, dispositivo de recuperaciA3n de Ambolos, dispositivo de captura de trombos, cesta vascular intervencionista, dispositivo de recuperaciA3n coronaria y perifArica
Dispositif de rAcupAration vasculaire en panier, panier vasculaire en nitinol, dispositif de rAcupAration d'emboles, dispositif de capture de thrombus, panier vasculaire interventionnel, dispositif de rAcupAration coronaire et pAriphArique
Ergonomic Design for Enhanced Control
The device boasts an ergonomic, non-slip handle that enables surgeons and interventionalists to maintain a secure and precise grip during complex procedures. This thoughtful design feature is critical for rigorous vascular interventions, especially when maneuvering through challenging, tortuous anatomy. Its lightweight and portable construction further enhance its ease-of-use and operational efficiency.
Advanced Material and Technology
Manufactured from a combination of medical-grade stainless steel and nitinol, the device exhibits superior flexibility and kink resistance. The minimally invasive endovascular technology not only ensures patient safety but also allows for real-time, silent operation. Its radiopaque tip and basket provide excellent fluoroscopic visibility, facilitating accurate placement and retrieval during procedures.
Comprehensive Safety and Compliance
Every vascular basket retrieval device is EO (Ethylene Oxide) sterilized, CE marked, and ISO 13485 certified, ensuring the highest standards of sterility and medical quality. Carefully packed in sterile blister packs, these devices offer a long shelf life of up to 5 years when stored in a cool, dry place. This commitment to safety and compliance underscores the product's reliability for clinical use in hospitals and advanced care centers.
FAQ's of Vascular Basket Retrieval Device:
Q: How is the vascular basket retrieval device used during procedures?
A: The device is manually operated and inserted via a compatible guide catheter (4 Fr-7 Fr) into the blood vessel. Once positioned near the target debris, the operator deploys the flexible basket under fluoroscopic guidance to capture and remove thrombus, calculi, or other foreign intravascular objects with precise control.Q: What are the main clinical benefits of using this device?
A: This retrieval device provides a minimally invasive solution for removing unwanted materials from blood vessels, reducing the need for open surgery, minimizing patient recovery time, and increasing the overall success rate due to its high flexibility and radiopaque visibility.Q: When should this device be stored or disposed of?
A: As a single-use, disposable instrument, it should be properly discarded after each procedure according to hospital protocols. Unused devices should be stored in a cool, dry place inside their original sterile blister packaging, with a maximum shelf life of five years from the date of manufacture.Q: Where can this device be used clinically?
A: It is suitable for use in a range of healthcare settings, including hospitals, surgical centers, and specialized cardiovascular units where endovascular or vascular retrieval procedures are performed.Q: What types of foreign bodies can this device retrieve?
A: The basket is designed to capture and retrieve a variety of intravascular debris, including thrombus (blood clots), calculi (stones), and other foreign bodies that may be obstructing blood vessels.Q: How does the device's radiopaque tip improve procedural accuracy?
A: The radiopaque tip and basket provide clear visibility under fluoroscopy, allowing the physician to precisely locate and manipulate the device for optimal foreign body capture and retrieval during the procedure.Q: What process ensures the sterility and compliance of this device?
A: Each device undergoes EO sterilization and rigorous quality checks in compliance with CE marking and ISO 13485 standards, and is individually packed in sterile blister packs to guarantee sterility until the point of use.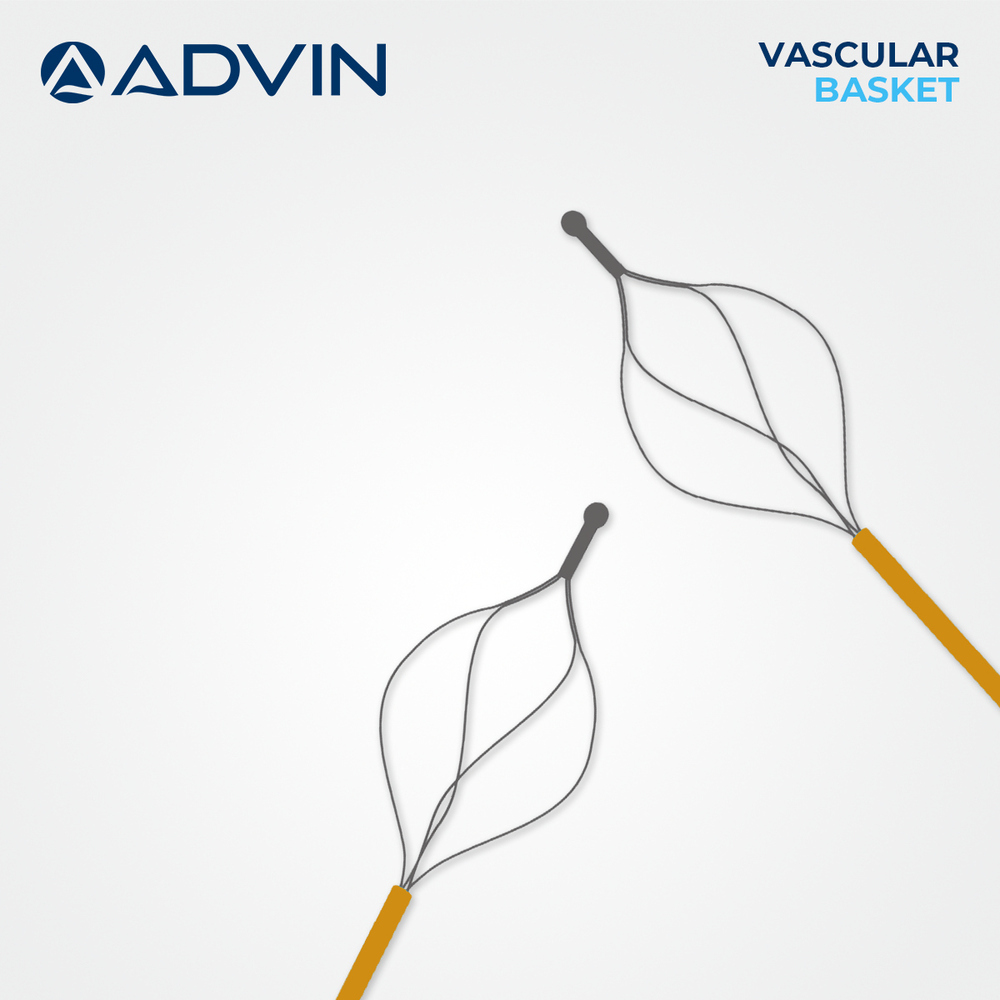



Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Cardiology Products Category
High Pressure Inflation Device
Price 20 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Storage Instructions : Store in a cool, dry place, away from direct sunlight
Technology : Manual inflation with pressure gauge
Wall Mounted : No, No
Noise Level : Silent/nonelectric
Balloon Tip Pacing Leads
Price 90 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Storage Instructions : Store in a Cool, Dry Place, Away from Direct Sunlight
Technology : Temporary Pacing Lead with Balloon Tip
Wall Mounted : No
Noise Level : Silent
Catheterization Transseptal Needle
Price 55 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Storage Instructions : Store in a Cool, Dry Place, Away from Direct Sunlight
Technology : Manual Percutaneous
Wall Mounted : No
Noise Level : Silent/None
Diagnostic Angiography Catheter
Price 7 USD ($) / Piece
Minimum Order Quantity : 1 Piece
Storage Instructions : Store in Cool, Dry Place
Technology : Softtip, Radioopaque Markers
Wall Mounted : No
Noise Level : Silent Operation

 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry


 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese